CGRP-CRLR/RAMP1 signal is important for stress-induced hematopoiesis
- PMID: 30674976
- PMCID: PMC6344543
- DOI: 10.1038/s41598-018-36796-0
CGRP-CRLR/RAMP1 signal is important for stress-induced hematopoiesis
Abstract
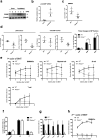
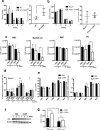
Ecotropic viral integration site-1 (EVI1) has a critical role in normal and malignant hematopoiesis. Since we previously identified high expression of calcitonin receptor like receptor (CRLR) in acute myeloid leukemia (AML) with high EVI1 expression, we here characterized the function of CRLR in hematopoiesis. Since higher expression of CRLR and receptor activity modifying protein 1 (RAMP1) was identified in immature hematopoietic bone marrow (BM) cells, we focused on calcitonin gene-related peptide (CGRP), a specific ligand for the CRLR/RAMP1 complex. To elucidate the role of CGRP in hematopoiesis, Ramp1-deficient (Ramp1-/-) mice were used. The steady-state hematopoiesis was almost maintained in Ramp1-/- mice; however, the BM repopulation capacity of Ramp1-/- mice was significantly decreased, and the transplanted Ramp1-/- BM mononuclear cells had low proliferation capacity with enhanced reactive oxygen species (ROS) production and cell apoptosis. Thus, CGRP is important for maintaining hematopoiesis during temporal exposures with proliferative stress. Moreover, continuous CGRP exposure to mice for two weeks induced a reduction in the number of BM immature hematopoietic cells along with differentiated myeloid cells. Since CGRP is known to be increased under inflammatory conditions to regulate immune responses, hematopoietic exhaustion by continuous CGRP secretion under chronic inflammatory conditions is probably one of the important mechanisms of anti-inflammatory responses.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Morrison SJ, Wandycz AM, Hemmati HD, Wright DE, Weissman IL. Identification of a lineage of multipotent hematopoietic progenitors. Development. 1997;124:1929–1939. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

