Coatings on mammalian cells: interfacing cells with their environment
- PMID: 30675178
- PMCID: PMC6337841
- DOI: 10.1186/s13036-018-0131-6
Coatings on mammalian cells: interfacing cells with their environment
Abstract
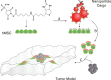
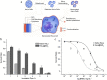
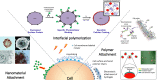
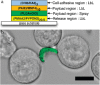
The research community is intent on harnessing increasingly complex biological building blocks. At present, cells represent a highly functional component for integration into higher order systems. In this review, we discuss the current application space for cellular coating technologies and emphasize the relationship between the target application and coating design. We also discuss how the cell and the coating interact in common analytical techniques, and where caution must be exercised in the interpretation of results. Finally, we look ahead at emerging application areas that are ideal for innovation in cellular coatings. In all, cellular coatings leverage the machinery unique to specific cell types, and the opportunities derived from these hybrid assemblies have yet to be fully realized.
Keywords: Cell therapy; Cellular coatings; Coatings; Mammalian cells; Polymers.
Conflict of interest statement
Not applicable.Not applicable.The authors declare that they have no competing interests.Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures












References
-
- Pathak CP, Sawhney AS, Hubbell JA. Rapid photopolymerization of immunoprotective gels in contact with cells and tissue. J Am Chem Soc. 1992;114:8311–8312. doi: 10.1021/ja00047a065. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

