CD4+ T cells promote humoral immunity and viral control during Zika virus infection
- PMID: 30677097
- PMCID: PMC6345435
- DOI: 10.1371/journal.ppat.1007474
CD4+ T cells promote humoral immunity and viral control during Zika virus infection
Erratum in
-
Correction: CD4+ T cells promote humoral immunity and viral control during Zika virus infection.PLoS Pathog. 2019 May 28;15(5):e1007821. doi: 10.1371/journal.ppat.1007821. eCollection 2019 May. PLoS Pathog. 2019. PMID: 31136625 Free PMC article.
Abstract
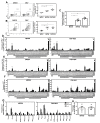
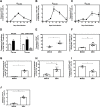
Several Zika virus (ZIKV) vaccines designed to elicit protective antibody (Ab) responses are currently under rapid development, but the underlying mechanisms that control the magnitude and quality of the Ab response remain unclear. Here, we investigated the CD4+ T cell response to primary intravenous and intravaginal infection with ZIKV. Using the LysMCre+Ifnar1fl/fl (myeloid type I IFN receptor-deficient) C57BL/6 mouse models, we identified six I-Ab-restricted ZIKV epitopes that stimulated CD4+ T cells with a predominantly cytotoxic Th1 phenotype in mice primed with ZIKV. Intravenous and intravaginal infection with ZIKV effectively induced follicular helper and regulatory CD4+ T cells. Treatment of mice with a CD4+ T cell-depleting Ab reduced the plasma cell, germinal center B cell, and IgG responses to ZIKV without affecting the CD8+ T cell response. CD4+ T cells were required to protect mice from a lethal dose of ZIKV after infection intravaginally, but not intravenously. However, adoptive transfer and peptide immunization experiments showed a role for memory CD4+ T cells in ZIKV clearance in mice challenged intravenously. These results demonstrate that CD4+ T cells are required mainly for the generation of a ZIKV-specific humoral response but not for an efficient CD8+ T cell response. Thus, CD4+ T cells could be important mediators of protection against ZIKV, depending on the infection or vaccination context.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
-
- Freour T, Mirallie S, Hubert B, Splingart C, Barriere P, Maquart M, et al. Sexual transmission of Zika virus in an entirely asymptomatic couple returning from a Zika epidemic area, France, April 2016. Euro Surveill. 2016;21(23). Epub 2016/06/18. 10.2807/1560-7917.ES.2016.21.23.30254 . - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

