Targeting the hepcidin-ferroportin pathway in anaemia of chronic kidney disease
- PMID: 30677788
- PMCID: PMC6475730
- DOI: 10.1111/bcp.13877
Targeting the hepcidin-ferroportin pathway in anaemia of chronic kidney disease
Abstract
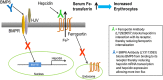
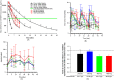
Aims: Erythropoiesis-stimulating agents used to treat anaemia in patients with chronic kidney disease (CKD) have been associated with cardiovascular adverse events. Hepcidin production, controlled by bone morphogenic protein 6 (BMP6), regulates iron homeostasis via interactions with the iron transporter, ferroportin. High hepcidin levels are thought to contribute to increased iron sequestration and subsequent anaemia in CKD patients. To investigate alternative therapies to erythropoiesis-stimulating agents for CKD patients, monoclonal antibodies, LY3113593 and LY2928057, targeting BMP6 and ferroportin respectively, were tested in CKD patients.
Methods: Preclinical in vitro/vivo data and clinical data in healthy subjects and CKD patients were used to illustrate the translation of pharmacological properties of LY3113593 and LY2928057, highlighting the novelty of targeting these nodes within the hepcidin-ferroportin pathway.
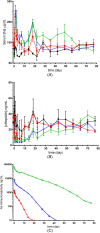
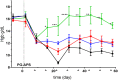
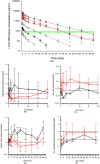
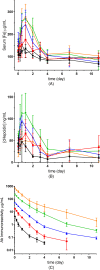
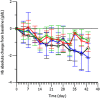
Results: LY2928057 bound ferroportin and blocked interactions with hepcidin, allowing iron efflux, leading to increased serum iron and transferrin saturation levels and increased hepcidin in monkeys and humans. In CKD patients, LY2928057 led to slower haemoglobin decline and reduction in ferritin (compared to placebo). Serum iron increase was (mean [90% confidence interval]) 1.98 [1.46-2.68] and 1.36 [1.22-1.51] fold-relative to baseline following LY2928057 600 mg and LY311593 150 mg respectively in CKD patients. LY3113593 specifically blocked BMP6 binding to its receptor and produced increases in iron and transferrin saturation and decreases in hepcidin preclinically and clinically. In CKD patients, LY3113593 produced an increase in haemoglobin and reduction in ferritin (compared to placebo).
Conclusion: LY3113593 and LY2928057 pharmacological effects (serum iron and ferritin) were translated from preclinical-to-clinical development. Such interventions may lead to new CKD anaemia treatments.
Trial registration: ClinicalTrials.gov NCT01330953 NCT01991483 NCT02144285 NCT02604160.
Keywords: chronic kidney disease; immunoglobulins; transport.
© 2019 The British Pharmacological Society.
Conflict of interest statement
D.R.W. is an employee and shareholder of Eli Lilly and Company and is the holder of the issued patent US 8183346,
Figures
Similar articles
-
Bone marrow iron distribution, hepcidin, and ferroportin expression in renal anemia.Hematology. 2015 Oct;20(9):543-52. doi: 10.1179/1607845415Y.0000000004. Epub 2015 Mar 8. Hematology. 2015. PMID: 25745821
-
Association between serum hepcidin-25 and primary resistance to erythropoiesis-stimulating agents in chronic kidney disease: a secondary analysis of the HERO trial.Nephrology (Carlton). 2017 Jul;22(7):548-554. doi: 10.1111/nep.12815. Nephrology (Carlton). 2017. PMID: 27171136 Clinical Trial.
-
Association between hepcidin, haemoglobin level and iron status in stage 4 chronic kidney disease patients with anaemia.J Pak Med Assoc. 2015 Apr;65(4):354-7. J Pak Med Assoc. 2015. PMID: 25976565
-
Targeting the hepcidin-ferroportin axis to develop new treatment strategies for anemia of chronic disease and anemia of inflammation.Am J Hematol. 2012 Apr;87(4):392-400. doi: 10.1002/ajh.23110. Epub 2012 Jan 31. Am J Hematol. 2012. PMID: 22290531 Free PMC article. Review.
-
Hepcidin.Adv Chronic Kidney Dis. 2019 Jul;26(4):298-305. doi: 10.1053/j.ackd.2019.04.005. Adv Chronic Kidney Dis. 2019. PMID: 31477260 Review.
Cited by
-
Interpreting Iron Homeostasis in Congenital and Acquired Disorders.Pharmaceuticals (Basel). 2023 Feb 21;16(3):329. doi: 10.3390/ph16030329. Pharmaceuticals (Basel). 2023. PMID: 36986429 Free PMC article. Review.
-
Iron and Chronic Kidney Disease: Still a Challenge.Front Med (Lausanne). 2020 Dec 18;7:565135. doi: 10.3389/fmed.2020.565135. eCollection 2020. Front Med (Lausanne). 2020. PMID: 33392212 Free PMC article. Review.
-
The Role of Iron in Benign and Malignant Hematopoiesis.Antioxid Redox Signal. 2021 Aug 20;35(6):415-432. doi: 10.1089/ars.2020.8155. Epub 2021 Jan 7. Antioxid Redox Signal. 2021. PMID: 33231101 Free PMC article. Review.
-
Treatment of Iron Deficiency Anemia in CKD and End-Stage Kidney Disease.Kidney Int Rep. 2021 Jun 5;6(9):2261-2269. doi: 10.1016/j.ekir.2021.05.020. eCollection 2021 Sep. Kidney Int Rep. 2021. PMID: 34514189 Free PMC article. Review.
-
Treatment of anemia in cancer patients undergoing chemotherapy with intravenous ferric carboxymaltose without erythropoiesis-stimulating agents.Ther Adv Med Oncol. 2020 Sep 9;12:1758835920953292. doi: 10.1177/1758835920953292. eCollection 2020. Ther Adv Med Oncol. 2020. PMID: 32952616 Free PMC article.
References
-
- Palmer SC, Navaneethan SD, Craig JC, et al. Meta‐analysis: erythropoiesis‐stimulating agents in patients with chronic kidney disease. Ann Intern Med. 2010;153(1):23‐33. - PubMed
-
- Drueke TB, Locatelli F, Clyne N, et al. Normalization of hemoglobin level in patients with chronic kidney disease and anemia. N Engl J Med. 2006;355(20):2071‐2084. - PubMed
-
- Fishbane S, Besarab A. Mechanism of increased mortality risk with erythropoietin treatment to higher hemoglobin targets. Clin J Am Soc Nephrol. 2007;2(6):1274‐1282. - PubMed
-
- Locatelli F, Del VL, Luise MC. Current and future chemical therapies for treating anaemia in chronic kidney disease. Expert Opin Pharmacother. 2017;18(8):781‐788. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

