miR-21 enhances the protective effect of loperamide on rat cardiomyocytes against hypoxia/reoxygenation, reactive oxygen species production and apoptosis via regulating Akap8 and Bard1 expression
- PMID: 30680008
- PMCID: PMC6327625
- DOI: 10.3892/etm.2018.7047
miR-21 enhances the protective effect of loperamide on rat cardiomyocytes against hypoxia/reoxygenation, reactive oxygen species production and apoptosis via regulating Akap8 and Bard1 expression
Abstract
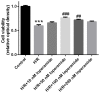
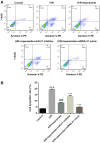
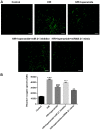
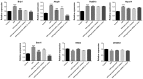
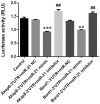
Effective therapies to reduce ischemia/reperfusion and hypoxia/reoxygenation injury are currently lacking. Furthermore, the effects of loperamide and microRNA (miR)-21 on hypoxia/reoxygenation injury of cardiomyocytes have remained to be elucidated. Therefore, the present study aimed to investigate the effect of loperamide and miR-21 on cardiomyocytes during hypoxia/reoxygenation injury, and to explore the underlying molecular mechanisms. H9c2 rat cardiomyocytes were pre-treated with loperamide prior to hypoxia/reoxygenation. The viability of H9c2 cells was measured with a cell counting kit 8 and apoptosis was detected with an Annexin V-phycoerythrin/7-aminoactinomycin D apoptosis kit. Furthermore, reactive oxygen species were detected with a specific kit. Genes regulated by miR-21 were screened with an mRNA chip and confirmed using reverse-transcription quantitative polymerase chain reaction analysis. The direct targeting relationship of miR-21 with certain mRNAs was then confirmed using a Dual-Luciferase Reporter Assay system. The results indicated that the apoptotic rate and reactive oxygen species levels in rat cardiomyocytes were markedly increased after hypoxia/reoxygenation treatment. Pre-treatment with loperamide significantly protected H9c2 cells against apoptosis and reactive oxygen species production after hypoxia/reoxygenation. The protection was markedly decreased by miR-21 inhibitor and enhanced by miR-21 mimics. Screening for genes associated with cardiomyocyte apoptosis revealed that the relative expression of A-kinase anchoring protein 8 (Akap8) and BRCA1 associated RING domain 1 (Bard1) was consistent with the experimental results on apoptosis and reactive oxygen species. Compared with the group treated by hypoxia/reoxygenation alone, pre-treatment with loperamide markedly decreased the expression of BRCA1-interacting protein C-terminal helicase 1, Akap8 and Bard1 after hypoxia/reoxygenation. The decrease in the expression of Akap8 and Bard1 was markedly attenuated by miR-21 inhibitor and enhanced by miR-21 mimics. miR-21 mimics directly targeted the 3'-untranslated region (UTR) of Akap8 and Bard1 mRNA to thereby decrease their expression. In conclusion, the protection of rat cardiomyocytes against hypoxia/reoxygenation-induced apoptosis and reactive oxygen species production by loperamide was markedly enhanced by miR-21. miR-21 directly targets the 3'-UTR of Akap8 and Bard1 mRNA and enhances the inhibitory effects of loperamide on Akap8 and Bard1 expression in rat cardiomyocytes after hypoxia/reoxygenation.
Keywords: cardiomyocytes; hypoxia; loperamide; miR-21; reoxygenation.
Figures

References
-
- O'Gara PT, Kushner FG, Ascheim DD, Casey DE, Jr, Chung MK, de Lemos JA, Ettinger SM, Fang JC, Fesmire FM, Franklin BA, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;127:e362–e425. - PubMed
-
- Fernández-Jiménez R, García-Prieto J, Sánchez-González J, Agüero J, López-Martín GJ, Galán-Arriola C, Molina-Iracheta A, Doohan R, Fuster V, Ibáñez B. Pathophysiology underlying the bimodal edema phenomenon after myocardial ischemia/reperfusion. J Am Coll Cardiol. 2015;66:816–828. doi: 10.1016/j.jacc.2015.06.023. - DOI - PubMed
-
- Bolli R, Becker L, Gross G, Mentzer R, Jr, Balshaw D, Lathrop DA. NHLBI Working Group on the Translation of Therapies for Protecting the Heart from Ischemia: Myocardial protection at a crossroads: The need for translation into clinical therapy. Circ Res. 2004;95:125–134. doi: 10.1161/01.RES.0000137171.97172.d7. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
