Interest to consider re-challenging by cetuximab and platinum containing regimen in recurrent Head and Neck Cancer
- PMID: 30680071
- PMCID: PMC6331031
- DOI: 10.18632/oncotarget.26506
Interest to consider re-challenging by cetuximab and platinum containing regimen in recurrent Head and Neck Cancer
Abstract
Background: The EXTREME protocol is the standard of care for recurrent or metastatic head and neck squamous-cell carcinoma (R/M HNSCC) in first line. Beyond the first-line except immunotherapy, poor efficacy was reported by second-line chemotherapy. Re-challenge strategies based on a repetition of the first line with platinum and cetuximab regimens might have been an option to consider.
Methods: We performed a retrospective study in order to assess the efficacy of the cetuximab plus platinum doublet-based chemotherapy regimen in patients with R/M HNSCC progressing after at least 3 months of cetuximab maintenance (EXTREME protocol). We complete a retrospective review of all medical records from R/M HNSCC patients treated after 16 weeks with the EXTREME regimen and treated with a re-challenge strategy between January 2010 and December 2014 in our institution (Centre Paul Strauss, Strasbourg, France).
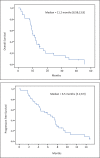
Results: 33 patients were identified. The re-challenged strategy provided an ORR in 33.3% of cases and a DCR of 69.6% of cases. The median OS and PFS observed from the second line were 11.2 months and 6.5 months for the subset re-challenged by EXTREME or PCC regimens respectively. The response rate between patients with a platin free interval within 3 and 6 months and greater than 6 months were equal. Drugs dose intensity were better with the PCC protocol than the EXTREME regimen used as a rechallenge.
Conclusions: This study suggest re-challenging strategy by these regimens could be considered beyond the first line as an option when the platin free interval is greater than 3 months.
Keywords: EXTREME; HNSCC; platinum free Interval; re-challenge.
Conflict of interest statement
CONFLICTS OF INTEREST CB has received honorariums for consulting by Merck, Astra-Zeneca, BMS, and has been Advisory board consultant for Merck and BMS. Other authors have no personal or institutional financial interest in any of the drugs, material, or devices described in this paper.
Figures
References
-
- Machiels JP, Haddad RI, Fayette J, Licitra LF, Tahara M, Vermorken JB, Clement PM, Gauler T, Cupissol D, Grau JJ, Guigay J, Caponigro F, de Castro G, Jr, et al. LUX-H&N 1 investigators Afatinib versus methotrexate as second-line treatment in patients with recurrent or metastatic squamous-cell carcinoma of the head and neck progressing on or after platinum-based therapy (LUX-Head & Neck 1): an open-label, randomised phase 3 trial. Lancet Oncol. 2015;16:583–94. doi: 10.1016/S1470-2045(15)70124-5. - DOI - PubMed
-
- Ferris RL, Blumenschein G, Jr, Fayette J, Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE, Even C, Worden F, Saba NF, Iglesias Docampo LC, et al. Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N Engl J Med. 2016;375:1856–67. doi: 10.1056/NEJMoa1602252. - DOI - PMC - PubMed
-
- Cohen EE, Machiels JP, Harrington KJ, Burtness B, Shin SW, Gause CK, Swift AM, Brown H, Perrone AM, Cheng JD, Swaby RF, Le Tourneau C. KEYNOTE-040: A phase III randomized trial of pembrolizumab (MK-3475) versus standard treatment in patients with recurrent or metastatic head and neck cancer. J Clin Oncol. 2015;33:TPS6084–6084.
LinkOut - more resources
Full Text Sources