Uptake and function of membrane-destabilizing cationic nanogels for intracellular drug delivery
- PMID: 30680315
- PMCID: PMC6336667
- DOI: 10.1002/btm2.10120
Uptake and function of membrane-destabilizing cationic nanogels for intracellular drug delivery
Abstract
The design of intracellular drug delivery vehicles demands an in-depth understanding of their internalization and function upon entering the cell to tailor the physicochemical characteristics of these platforms and achieve efficacious treatments. Polymeric cationic systems have been broadly accepted to be membrane disruptive thus being beneficial for drug delivery inside the cell. However, if excessive destabilization takes place, it can lead to adverse effects. One of the strategies used to modulate the cationic charge is the incorporation of hydrophobic moieties, thus increasing the hydrophobic content. We have demonstrated the successful synthesis of nanogels based on diethylaminoethyl methacrylate and poly(ethylene glycol) methyl ether methacrylate. Addition of the hydrophobic monomers tert-butyl methacrylate or 2-(tert-butylamino)ethyl methacrylate shows improved polymer hydrophobicity and modulation of the critical swelling pH. Here, we evaluate the cytocompatibility, uptake, and function of these membrane-destabilizing cationic methacrylated nanogels using in vitro models. The obtained results suggest that the incorporation of hydrophobic monomers decreases the cytotoxicity of the nanogels to epithelial colorectal adenocarcinoma cells. Furthermore, analysis of the internalization pathways of these vehicles using inhibitors and imaging flow cytometry showed a significant decrease in uptake when macropinocytosis/phagocytosis inhibitors were present. The membrane-disruptive abilities of the cationic polymeric nanogels were confirmed using three different models. They demonstrated to cause hemolysis in sheep erythrocytes, lactate dehydrogenase leakage from a model cell line, and disrupt giant unilamellar vesicles. These findings provide new insights of the potential of polymeric nanoformulations for intracellular delivery.
Keywords: cationic; drug delivery; intracellular; nanoparticles; polymer.
Figures
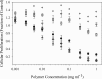
 ), PDETB20 (
), PDETB20 ( ), PDETB30 (
), PDETB30 ( ), PDETBA20 (
), PDETBA20 ( ), or PDETBA30 (
), or PDETBA30 ( ). Proliferation of Caco‐2 cells was determined by MTS assay following 90 min nanogel exposure and is expressed as a fraction of the control (untreated) cells. Data are expressed as mean ± SEM, n = 8. Statistical significance determined via pairwise t‐test between cells exposed to PDETB20 and PDET or PDETB30 and PDET (*p < .005)
). Proliferation of Caco‐2 cells was determined by MTS assay following 90 min nanogel exposure and is expressed as a fraction of the control (untreated) cells. Data are expressed as mean ± SEM, n = 8. Statistical significance determined via pairwise t‐test between cells exposed to PDETB20 and PDET or PDETB30 and PDET (*p < .005)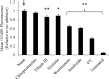
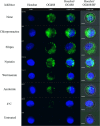

 ), and PDETBA30 (
), and PDETBA30 ( ) in 150 mM phosphate buffer at early endosomal pH (pH 6.0). Erythrocytes exposed to various polymer concentrations for 60 min at 37 °C. Data points represent the mean of triplicate samples ± SD. Panels (c) and (d) show the influence of TBMA incorporation on pyrene excitation (I338/I333 ratio) in P(DEAEMA‐co‐TBMA‐g‐PEGMA) nanogels (panel c), and of the inclusion of TBAEMA in the nanogel formulation (panel d). Nanogels suspended at 0.5 mg ml−1 and pyrene dissolved at 6 × 10–7 M in 100 mM phosphate buffers at designated pH values. Panel (e) shows a summary table of the pH transition (pHapp) of cationic nanogel formulations
) in 150 mM phosphate buffer at early endosomal pH (pH 6.0). Erythrocytes exposed to various polymer concentrations for 60 min at 37 °C. Data points represent the mean of triplicate samples ± SD. Panels (c) and (d) show the influence of TBMA incorporation on pyrene excitation (I338/I333 ratio) in P(DEAEMA‐co‐TBMA‐g‐PEGMA) nanogels (panel c), and of the inclusion of TBAEMA in the nanogel formulation (panel d). Nanogels suspended at 0.5 mg ml−1 and pyrene dissolved at 6 × 10–7 M in 100 mM phosphate buffers at designated pH values. Panel (e) shows a summary table of the pH transition (pHapp) of cationic nanogel formulations
 ) exposure to PDET (a), PDETB30 (b), and PDETBA30 (c). Data points represent the sample mean ± SEM (n = 4). LDH leakage calculated relative to untreated cells and surfactant‐lysed cells
) exposure to PDET (a), PDETB30 (b), and PDETBA30 (c). Data points represent the sample mean ± SEM (n = 4). LDH leakage calculated relative to untreated cells and surfactant‐lysed cells
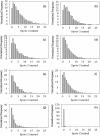
 ), PDETB10 (
), PDETB10 ( ), PDETB20 (
), PDETB20 ( ), or PDETB30 (
), or PDETB30 ( ) for 60 min (a) or PDET (
) for 60 min (a) or PDET ( ), PDETBA10 (
), PDETBA10 ( ), PDETBA20 (
), PDETBA20 ( ), or PDETBA30 (
), or PDETBA30 ( ) for 60 min (b)
) for 60 min (b)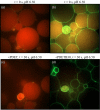
References
-
- Szachowicz‐Petelska B, Figaszewski Z, Lewandowski W. Mechanisms of transport across cell membranes of complexes contained in antitumour drugs. Int J Pharm. 2001;222(2):169‐182. - PubMed
-
- Desai MP, Labhasetwar V, Walter E, Levy RJ, Amidon GL. The mechanism of uptake of biodegradable microparticles in Caco‐2 cells is size dependent. Pharm Res. 1997;14(11):1568‐1573. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

