Side-Chain Pruning Has Limited Impact on Substrate Preference in a Promiscuous Enzyme
- PMID: 30687578
- PMCID: PMC6345240
- DOI: 10.1021/acscatal.8b03793
Side-Chain Pruning Has Limited Impact on Substrate Preference in a Promiscuous Enzyme
Abstract
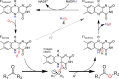
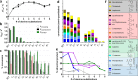
Detoxifying enzymes such as flavin-containing monooxygenases deal with a huge array of highly diverse xenobiotics and toxic compounds. In addition to being of high physiological relevance, these drug-metabolizing enzymes are useful catalysts for synthetic chemistry. Despite the wealth of studies, the molecular basis of their relaxed substrate selectivity remains an open question. Here, we addressed this issue by applying a cumulative alanine mutagenesis approach to cyclohexanone monooxygenase from Thermocrispum municipale, a flavin-dependent Baeyer-Villiger monooxygenase which we chose as a model system because of its pronounced thermostability and substrate promiscuity. Simultaneous removal of up to eight noncatalytic active-site side chains including four phenylalanines had no effect on protein folding, thermostability, and cofactor loading. We observed a linear decrease in activity, rather than a selectivity switch, and attributed this to a less efficient catalytic environment in the enlarged active-site space. Time-resolved kinetic studies confirmed this interpretation. We also determined the crystal structure of the enzyme in complex with a mimic of the reaction intermediate that shows an unaltered overall protein conformation. These findings led us to propose that this cyclohexanone monooxygenase may lack a distinct substrate selection mechanism altogether. We speculate that the main or exclusive function of the protein shell in promiscuous enzymes might be the stabilization and accessibility of their very reactive catalytic intermediates.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Exploiting Cofactor Versatility to Convert a FAD-Dependent Baeyer-Villiger Monooxygenase into a Ketoreductase.Angew Chem Int Ed Engl. 2019 Oct 7;58(41):14499-14503. doi: 10.1002/anie.201907606. Epub 2019 Sep 5. Angew Chem Int Ed Engl. 2019. PMID: 31423719
-
Enantioselective sulfoxidations employing the thermostable cyclohexanone monooxygenase from Thermocrispum municipale.Enzyme Microb Technol. 2018 Jun;113:24-28. doi: 10.1016/j.enzmictec.2018.02.006. Epub 2018 Feb 21. Enzyme Microb Technol. 2018. PMID: 29602383
-
Application of a thermostable Baeyer-Villiger monooxygenase for the synthesis of branched polyester precursors.J Chem Technol Biotechnol. 2018 Aug;93(8):2131-2140. doi: 10.1002/jctb.5623. Epub 2018 Apr 16. J Chem Technol Biotechnol. 2018. PMID: 30069077 Free PMC article.
-
Toward Upscaled Biocatalytic Preparation of Lactone Building Blocks for Polymer Applications.Org Process Res Dev. 2018 Jul 20;22(7):803-812. doi: 10.1021/acs.oprd.8b00079. Epub 2018 Jun 12. Org Process Res Dev. 2018. PMID: 30271110 Free PMC article. Review.
-
[Baeyer-Villiger monooxygenases in the biosynthesis of microbial secondary metabolites].Sheng Wu Gong Cheng Xue Bao. 2019 Mar 25;35(3):351-362. doi: 10.13345/j.cjb.180294. Sheng Wu Gong Cheng Xue Bao. 2019. PMID: 30912344 Review. Chinese.
Cited by
-
Bioinformatic Mining and Structure-Activity Profiling of Baeyer-Villiger Monooxygenases from Mycobacterium tuberculosis.mSphere. 2022 Apr 27;7(2):e0048221. doi: 10.1128/msphere.00482-21. Epub 2022 Mar 17. mSphere. 2022. PMID: 35296143 Free PMC article.
-
Stabilization of cyclohexanone monooxygenase by computational and experimental library design.Biotechnol Bioeng. 2019 Sep;116(9):2167-2177. doi: 10.1002/bit.27022. Epub 2019 Jun 24. Biotechnol Bioeng. 2019. PMID: 31124128 Free PMC article.
-
Mechanisms of promiscuity among drug metabolizing enzymes and drug transporters.FEBS J. 2020 Apr;287(7):1306-1322. doi: 10.1111/febs.15116. Epub 2019 Nov 12. FEBS J. 2020. PMID: 31663687 Free PMC article. Review.
References
-
- Iyanagi T.Molecular Mechanism of Phase I and Phase II Drug-Metabolizing Enzymes: Implications for Detoxification. In International Review of Cytology; Academic Press: New York, 2007; pp 35–112. - PubMed
-
- Guengerich F. P.Human Cytochrome P450 Enzymes. In Cytochrome P450; Springer: Berlin, 2005; pp 377–530.
LinkOut - more resources
Full Text Sources
