The Cutaneous Inflammatory Response to Thermal Burn Injury in a Murine Model
- PMID: 30696002
- PMCID: PMC6387172
- DOI: 10.3390/ijms20030538
The Cutaneous Inflammatory Response to Thermal Burn Injury in a Murine Model
Abstract
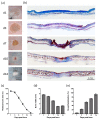
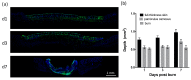
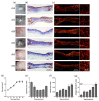
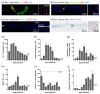
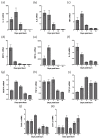
Many burn interventions aim to target the inflammatory response as a means of enhancing healing or limiting hypertrophic scarring. Murine models of human burns have been developed, but the inflammatory response to injury in these models has not been well defined. The aim of this study was to profile inflammatory cell populations and gene expression relative to healing and scarring in a murine model of thermal burns. Cutaneous injuries were created on the dorsal region of C57Bl/6 mice using a heated metal rod. Animals were euthanized at selected time points over ten weeks, with the lesions evaluated using macroscopic measurements, histology, immunofluorescent histochemistry and quantitative PCR. The burn method generated a reproducible, partial-thickness injury that healed within two weeks through both contraction and re-epithelialization, in a manner similar to human burns. The injury caused an immediate increase in pro-inflammatory cytokine and chemokine expression, coinciding with an influx of neutrophils, and the disappearance of Langerhans cells and mast cells. This preceded an influx of dendritic cells and macrophages, a quarter of which displayed an inflammatory (M1) phenotype, with both populations peaking at closure. As with human burns, the residual scar increased in size, epidermal and dermal thickness, and mast cell numbers over 10 weeks, but abnormal collagen I-collagen III ratios, fibre organization and macrophage populations resolved 3⁻4 weeks after closure. Characterisation of the inflammatory response in this promising murine burn model will assist future studies of burn complications and aid in the preclinical testing of new anti-inflammatory and anti-scarring therapies.
Keywords: Langerhans cell; collagen; dendritic cell; hypertrophic scar; inflammation; macrophage; mast cell; mice; neutrophil; thermal burn.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
-
- WHO Fact Sheet: Burns. [(accessed on 6 March 2018)]; Available online: https://www.who.int/news-room/fact-sheets/detail/burns.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

