Astrocyte activation in the periaqueductal gray promotes descending facilitation to cancer-induced bone pain through the JNK MAPK signaling pathway
- PMID: 30700204
- PMCID: PMC6388461
- DOI: 10.1177/1744806919831909
Astrocyte activation in the periaqueductal gray promotes descending facilitation to cancer-induced bone pain through the JNK MAPK signaling pathway
Abstract
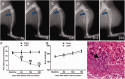
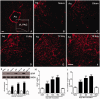
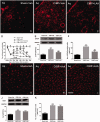
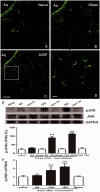
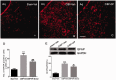
Descending nociceptive modulation from the supraspinal structures has an important role in cancer-induced bone pain (CIBP). Midbrain ventrolateral periaqueductal gray (vlPAG) is a critical component of descending nociceptive circuits; nevertheless, its precise cellular and molecular mechanisms involved in descending facilitation remain elusive. Our previous study has shown that the activation of p38 MAPK in vlPAG microglia is essential for the neuropathic pain sensitization. However, the existence of potential connection between astrocytes and c-Jun N-terminal kinase (JNK) pathway in CIBP has not yet been elucidated. The following study examines the involvement of astrocyte activation and upregulation of p-JNK in vlPAG, using a CIBP rat model. Briefly, CIBP was mimicked by an intramedullary injection of Walker 256 mammary gland carcinoma cells into the animal tibia. A significant increase in expression levels of astrocytes in the vlPAG of CIBP rats was observed. Furthermore, stereotaxic microinjection of the astrocytic cytotoxin L-α-aminoadipic acid decreased the mechanical allodynia as well as established and reversed the astrocyte activation in CIBP rats. A significant increase in expression levels of p-JNK in astrocytes in vlPAG of CIBP rats was also observed. Moreover, the intrathecal administration of JNK inhibitors SP600125 reduced the expression of glial fibrillary acidic protein, while microinjection of the SP600125 decreased the mechanical allodynia of CIBP rats. These results suggested that CIBP is associated with astrocyte activation in the vlPAG that probably participates in driving descending pain facilitation through the JNK MAPK signaling pathway. To sum up, these findings reveal a novel site of astrocytes modulation of CIBP.
Keywords: Cancer-induced bone pain; astrocyte; c-Jun N-terminal kinase; descending facilitation; hyperalgesia; periaqueductal gray.
Figures


Similar articles
-
Glial activation in the periaqueductal gray promotes descending facilitation of neuropathic pain through the p38 MAPK signaling pathway.J Neurosci Res. 2016 Jan;94(1):50-61. doi: 10.1002/jnr.23672. Epub 2015 Oct 1. J Neurosci Res. 2016. PMID: 26423029
-
Crosstalk between NFκB-dependent astrocytic CXCL1 and neuron CXCR2 plays a role in descending pain facilitation.J Neuroinflammation. 2019 Jan 3;16(1):1. doi: 10.1186/s12974-018-1391-2. J Neuroinflammation. 2019. PMID: 30606213 Free PMC article.
-
The Role of Spinal GABAB Receptors in Cancer-Induced Bone Pain in Rats.J Pain. 2017 Aug;18(8):933-946. doi: 10.1016/j.jpain.2017.02.438. Epub 2017 Mar 18. J Pain. 2017. PMID: 28323246
-
Activation of JNK pathway in persistent pain.Neurosci Lett. 2008 Jun 6;437(3):180-3. doi: 10.1016/j.neulet.2008.03.017. Epub 2008 Mar 13. Neurosci Lett. 2008. PMID: 18455869 Free PMC article. Review.
-
Cellular and circuit diversity determines the impact of endogenous opioids in the descending pain modulatory pathway.Front Syst Neurosci. 2022 Aug 15;16:963812. doi: 10.3389/fnsys.2022.963812. eCollection 2022. Front Syst Neurosci. 2022. PMID: 36045708 Free PMC article. Review.
Cited by
-
Rosiglitazone Alleviates Mechanical Allodynia of Rats with Bone Cancer Pain through the Activation of PPAR-γ to Inhibit the NF-κB/NLRP3 Inflammatory Axis in Spinal Cord Neurons.PPAR Res. 2021 Aug 25;2021:6086265. doi: 10.1155/2021/6086265. eCollection 2021. PPAR Res. 2021. PMID: 34484316 Free PMC article.
-
Astrocytic Regulation of Neural Circuits Underlying Behaviors.Cells. 2021 Feb 1;10(2):296. doi: 10.3390/cells10020296. Cells. 2021. PMID: 33535587 Free PMC article. Review.
-
Macrophages and glial cells: Innate immune drivers of inflammatory arthritic pain perception from peripheral joints to the central nervous system.Front Pain Res (Lausanne). 2022 Oct 26;3:1018800. doi: 10.3389/fpain.2022.1018800. eCollection 2022. Front Pain Res (Lausanne). 2022. PMID: 36387416 Free PMC article. Review.
-
Astrocytes in chronic pain and itch.Nat Rev Neurosci. 2019 Nov;20(11):667-685. doi: 10.1038/s41583-019-0218-1. Epub 2019 Sep 19. Nat Rev Neurosci. 2019. PMID: 31537912 Free PMC article. Review.
-
Inhibiting the JNK Signaling Pathway Attenuates Hypersensitivity and Anxiety-Like Behavior in a Rat Model of Non-specific Chronic Low Back Pain.J Mol Neurosci. 2024 Jul 24;74(3):73. doi: 10.1007/s12031-024-02252-0. J Mol Neurosci. 2024. PMID: 39046556
References
-
- van den Beuken-van Everdingen MHJ, de Rijke JM, Kessels AG, Schouten HC, van Kleef M, Patijn J. Prevalence of pain in patients with cancer: a systematic review of the past 40 years. Ann Oncol 2007; 18: 1437–1449. - PubMed
-
- Goblirsch MJ, Zwolak P, Clohisy DR. Advances in understanding bone cancer pain. J Cell Biochem 2005; 96: 682–688. - PubMed
-
- Falk S, Dickenson AH. Pain and nociception: mechanisms of cancer-induced bone pain. JCO 2014; 32: 1647–1654. - PubMed
-
- Dubovy P, Klusáková I, Hradilova-Svizenska I, Joukal M, Boadas-Vaello P. Activation of astrocytes and microglial cells and CCL2/CCR2 upregulation in the dorsolateral and ventrolateral nuclei of periaqueductal gray and rostral ventromedial medulla following different types of sciatic nerve injury. Front Cell Neurosci 2018; 12: 40. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

