EGFR-Aurka Signaling Rescues Polarity and Regeneration Defects in Dystrophin-Deficient Muscle Stem Cells by Increasing Asymmetric Divisions
- PMID: 30713094
- PMCID: PMC6408300
- DOI: 10.1016/j.stem.2019.01.002
EGFR-Aurka Signaling Rescues Polarity and Regeneration Defects in Dystrophin-Deficient Muscle Stem Cells by Increasing Asymmetric Divisions
Abstract
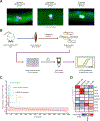
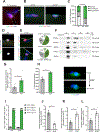
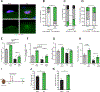
Loss of dystrophin expression in Duchenne muscular dystrophy (DMD) causes progressive degeneration of skeletal muscle, which is exacerbated by reduced self-renewing asymmetric divisions of muscle satellite cells. This, in turn, affects the production of myogenic precursors and impairs regeneration and suggests that increasing such divisions may be beneficial. Here, through a small-molecule screen, we identified epidermal growth factor receptor (EGFR) and Aurora kinase A (Aurka) as regulators of asymmetric satellite cell divisions. Inhibiting EGFR causes a substantial shift from asymmetric to symmetric division modes, whereas EGF treatment increases asymmetric divisions. EGFR activation acts through Aurka to orient mitotic centrosomes, and inhibiting Aurka blocks EGF stimulation-induced asymmetric division. In vivo EGF treatment markedly activates asymmetric divisions of dystrophin-deficient satellite cells in mdx mice, increasing progenitor numbers, enhancing regeneration, and restoring muscle strength. Therefore, activating an EGFR-dependent polarity pathway promotes functional rescue of dystrophin-deficient satellite cells and enhances muscle force generation.
Keywords: Aurka; Duchenne muscular dystrophy; EGF; EGFR; apicobasal polarity; asymmetric cell division; muscle stem cell; satellite cell; skeletal muscle.
Copyright © 2019 Elsevier Inc. All rights reserved.
Conflict of interest statement
CONFLICTS OF INTEREST
M.A.R. is a Founding Scientist of Satellos Bioscience Inc. The other authors declare no competing financial interests.
Figures
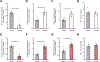


Similar articles
-
Dystrophin expression in muscle stem cells regulates their polarity and asymmetric division.Nat Med. 2015 Dec;21(12):1455-63. doi: 10.1038/nm.3990. Epub 2015 Nov 16. Nat Med. 2015. PMID: 26569381 Free PMC article.
-
Intrinsic Muscle Stem Cell Dysfunction Contributes to Impaired Regeneration in the mdx Mouse.J Cachexia Sarcopenia Muscle. 2025 Feb;16(1):e13682. doi: 10.1002/jcsm.13682. J Cachexia Sarcopenia Muscle. 2025. PMID: 39723578 Free PMC article.
-
Unacylated Ghrelin Enhances Satellite Cell Function and Relieves the Dystrophic Phenotype in Duchenne Muscular Dystrophy mdx Model.Stem Cells. 2017 Jul;35(7):1733-1746. doi: 10.1002/stem.2632. Epub 2017 May 7. Stem Cells. 2017. PMID: 28436144
-
Satellite Cells in Muscular Dystrophy - Lost in Polarity.Trends Mol Med. 2016 Jun;22(6):479-496. doi: 10.1016/j.molmed.2016.04.002. Epub 2016 May 5. Trends Mol Med. 2016. PMID: 27161598 Free PMC article. Review.
-
How does dystrophin deficiency lead to muscle degeneration?--evidence from the mdx mouse.Neuromuscul Disord. 1995 Nov;5(6):445-56. doi: 10.1016/0960-8966(95)00001-4. Neuromuscul Disord. 1995. PMID: 8580726 Review.
Cited by
-
A reference single-cell transcriptomic atlas of human skeletal muscle tissue reveals bifurcated muscle stem cell populations.Skelet Muscle. 2020 Jul 6;10(1):19. doi: 10.1186/s13395-020-00236-3. Skelet Muscle. 2020. PMID: 32624006 Free PMC article.
-
Sertoli Cells Improve Myogenic Differentiation, Reduce Fibrogenic Markers, and Induce Utrophin Expression in Human DMD Myoblasts.Biomolecules. 2021 Oct 12;11(10):1504. doi: 10.3390/biom11101504. Biomolecules. 2021. PMID: 34680138 Free PMC article.
-
Loss of full-length dystrophin expression results in major cell-autonomous abnormalities in proliferating myoblasts.Elife. 2022 Sep 27;11:e75521. doi: 10.7554/eLife.75521. Elife. 2022. PMID: 36164827 Free PMC article.
-
HDAC inhibitors as pharmacological treatment for Duchenne muscular dystrophy: a discovery journey from bench to patients.Trends Mol Med. 2024 Mar;30(3):278-294. doi: 10.1016/j.molmed.2024.01.007. Epub 2024 Feb 26. Trends Mol Med. 2024. PMID: 38408879 Free PMC article. Review.
-
Aptamer-conjugated gold nanoparticles enable oligonucleotide delivery into muscle stem cells to promote regeneration of dystrophic muscles.Nat Commun. 2025 Jan 10;16(1):577. doi: 10.1038/s41467-024-55223-9. Nat Commun. 2025. PMID: 39794309 Free PMC article.
References
-
- Anderson MS, and Kunkel LM (1992). The molecular and biochemical basis of Duchenne muscular dystrophy. Trends Biochem. Sci 17, 289–292. - PubMed
-
- Ando R, Ikegami H, Sakiyama M, Ooike S, Hayashi M, Fujino Y, Abe D, Nakamura H, Mishina T, Kato H, et al. (2010). 3-Cyano-6-(5-methyl-3-pyrazoloamino)pyridines: selective Aurora A kinase inhibitors. Bioorg. Med. Chem. Lett 20, 4709–4711. - PubMed
-
- Bell CD, and Conen PE (1968). Histopathological changes in Duchenne muscular dystrophy. J. Neurol. Sci 7, 529–544. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

