Structure Elucidation and Biological Evaluation of Maitotoxin-3, a Homologue of Gambierone, from Gambierdiscus belizeanus
- PMID: 30717108
- PMCID: PMC6409949
- DOI: 10.3390/toxins11020079
Structure Elucidation and Biological Evaluation of Maitotoxin-3, a Homologue of Gambierone, from Gambierdiscus belizeanus
Abstract
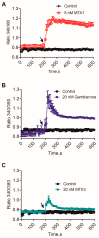
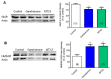
Gambierdiscus species are the producers of the marine toxins ciguatoxins and maitotoxins which cause worldwide human intoxications recognized as Ciguatera Fish Poisoning. A deep chemical investigation of a cultured strain of G. belizeanus, collected in the Caribbean Sea, led to the identification of a structural homologue of the recently described gambierone isolated from the same strain. The structure was elucidated mainly by comparison of NMR and MS data with those of gambierone and ascertained by 2D NMR data analyses. Gratifyingly, a close inspection of the MS data of the new 44-methylgambierone suggests that this toxin would actually correspond to the structure of maitotoxin-3 (MTX3, m/z 1039.4957 for the protonated adduct) detected in 1994 in a Pacific strain of Gambierdiscus and recently shown in routine monitoring programs. Therefore, this work provides for the first time the chemical identification of the MTX3 molecule by NMR. Furthermore, biological data confirmed the similar activities of both gambierone and 44-methylgambierone. Both gambierone and MTX3 induced a small increase in the cytosolic calcium concentration but only MTX3 caused cell cytotoxicity at micromolar concentrations. Moreover, chronic exposure of human cortical neurons to either gambierone or MTX3 altered the expression of ionotropic glutamate receptors, an effect already described before for the synthetic ciguatoxin CTX3C. However, even when gambierone and MTX3 affected glutamate receptor expression in a similar manner their effect on receptor expression differed from that of CTX3C, since both toxins decreased AMPA receptor levels while increasing N-methyl-d-aspartate (NMDA) receptor protein. Thus, further studies should be pursued to clarify the similarities and differences in the biological activity between the known ciguatoxins and the new identified molecule as well as its contribution to the neurological symptoms of ciguatera.
Keywords: 44-methylgambierone; Gambierdiscus belizeanus; ciguatera; cytosolic calcium concentration; gambierone; glutamate receptors; maitotoxin; maitotoxin-3; neurotoxicity.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
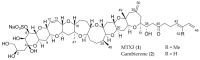


References
-
- Friedman M.A., Fernandez M., Backer L.C., Dickey R.W., Bernstein J., Schrank K., Kibler S., Stephan W., Gribble M.O., Bienfang P., et al. An Updated Review of Ciguatera Fish Poisoning: Clinical, Epidemiological, Environmental, and Public Health Management. Mar. Drugs. 2017;15:72. doi: 10.3390/md15030072. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

