Synthesis, Characterization, and Modification of Alumina Nanoparticles for Cationic Dye Removal
- PMID: 30717156
- PMCID: PMC6384569
- DOI: 10.3390/ma12030450
Synthesis, Characterization, and Modification of Alumina Nanoparticles for Cationic Dye Removal
Abstract
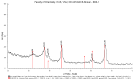
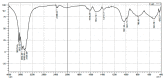
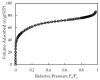
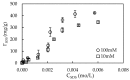
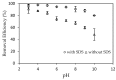
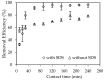
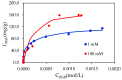
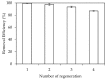
In the present study, alumina nanoparticles (nano-alumina) which were successfully fabricated by solvothermal method, were characterized by X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), Transmission Electron Microscopy (TEM), and Brunauer⁻Emmett⁻Teller (BET) methods. The removal of cationic dye, Rhodamine B (RhB), through adsorption method using synthesized nano-alumina with surface modification by anionic surfactant was also investigated. An anionic surfactant, sodium dodecyl sulfate (SDS) was used to modify nano-alumina surface at low pH and high ionic strength increased the removal efficiency of RhB significantly. The optimum adsorption conditions of contact time, pH, and adsorbent dosage for RhB removal using SDS modified nano-alumina (SMNA) were found to be 120 min, pH 4, and 5 mg/mL respectively. The RhB removal using SMNA reached a very high removal efficiency of 100%. After four times regeneration of adsorbent, the removal efficiency of RhB using SMNA was still higher than 86%. Adsorption isotherms of RhB onto SMNA at different salt concentrations were fitted well by a two-step model. A very high adsorption capacity of RhB onto SMNA of 165 mg/g was achieved. Adsorption mechanisms of RhB onto SMNA were discussed on the basis of the changes in surface modifications, the change in surface charges and adsorption isotherms.
Keywords: Adsorption isotherm; Alumina nanoparticles; Rhodamine B; SDS; Two-step model.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Adak A., Bandyopadhyay M., Pal A. Removal of crystal violet dye from wastewater by surfactant-modified alumina. Sep. Purif. Technol. 2005;44:139–144. doi: 10.1016/j.seppur.2005.01.002. - DOI
-
- Wong Y.C., Szeto Y.S., Cheung W.H., McKay G. Equilibrium Studies for Acid Dye Adsorption onto Chitosan. Langmuir. 2003;19:7888–7894. doi: 10.1021/la030064y. - DOI
LinkOut - more resources
Full Text Sources

