Synthesis of Catalytic Nanoporous Metallic Thin Films on Polymer Membranes
- PMID: 30718940
- PMCID: PMC6358282
- DOI: 10.1021/acs.iecr.8b00053
Synthesis of Catalytic Nanoporous Metallic Thin Films on Polymer Membranes
Abstract
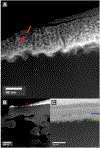
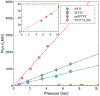
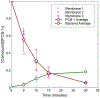
Composite membranes were produced with a metallic thin film forming the upper layer of the composite on a porous polymer support. Commercially available membranes were used as supports with both micron and nanometer scale pores. Alloy films of ~110 nm thickness were deposited via magnetron sputtering to produce the top layer of the composite. Dealloying the film with sulfuric acid allowed the creation of a nanoporous film structure with a ligament size of 7.7 ± 2.5 nm. Resulting composite membranes were permeable to water at all stages of production, and a UF PSf membrane with 90 nm of nanoporous Fe/Pd on top showed a flux of 183 LHM/bar. The films were evaluated for dechlorination of toxic polychlorinated biphenyls from water. At a loading of 6.6 mg/L of Pd attached to 13.2 cm2 support in a 2.5 ppm PCB-1 solution with 1.5 ppm dissolved H2, over 90% of PCB-1 was removed from solution in 30 minutes, which produced the expected product biphenyl from the dechlorination reaction.
Keywords: composite membranes; dealloying; dechlorination; nanoporous metals.
Figures





References
-
- Werber JR; Osuji CO; Elimelech M Materials for next-generation desalination and water purification membranes. Nat. Rev. Mater. 2016, 1, 16018.
-
- Raney M Method of Preparing Catalytic Material. USPO 1,563,587, 1925.
-
- Fouilloux P The nature of raney nickel, its adsorbed hydrogen and its catalytic activity for hydrogenation reactions (review). Appl. Catal. 1983, 8 (1), 1–42.
-
- Erlebacher J; Aziz MJ; Karma A; Dimitrov N; Sieradzki K Evolution of nanoporosity in dealloying. Nature 2001, 410 (6827), 450–453. - PubMed
-
- Erlebacher J An Atomistic Description of Dealloying Porosity Evolution, the Critical Potential, and Rate-Limiting Behavior. J. Electrochem. Soc. 2004, 151 (10), 10.
Grants and funding
LinkOut - more resources
Full Text Sources
