Genetic lesioning of histamine neurons increases sleep-wake fragmentation and reveals their contribution to modafinil-induced wakefulness
- PMID: 30722053
- PMCID: PMC6519916
- DOI: 10.1093/sleep/zsz031
Genetic lesioning of histamine neurons increases sleep-wake fragmentation and reveals their contribution to modafinil-induced wakefulness
Erratum in
-
Corrigendum: Genetic lesioning of histamine neurons increases sleep-wake fragmentation and reveals their contribution to modafinil-induced wakefulness.Sleep. 2019 Aug 1;42(8):zsz107. doi: 10.1093/sleep/zsz107. Sleep. 2019. PMID: 31152538 Free PMC article. No abstract available.
Abstract
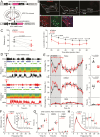
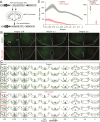
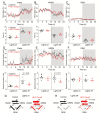
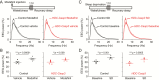
Acute chemogenetic inhibition of histamine (HA) neurons in adult mice induced nonrapid eye movement (NREM) sleep with an increased delta power. By contrast, selective genetic lesioning of HA neurons with caspase in adult mice exhibited a normal sleep-wake cycle overall, except at the diurnal start of the lights-off period, when they remained sleepier. The amount of time spent in NREM sleep and in the wake state in mice with lesioned HA neurons was unchanged over 24 hr, but the sleep-wake cycle was more fragmented. Both the delayed increase in wakefulness at the start of the night and the sleep-wake fragmentation are similar phenotypes to histidine decarboxylase knockout mice, which cannot synthesize HA. Chronic loss of HA neurons did not affect sleep homeostasis after sleep deprivation. However, the chronic loss of HA neurons or chemogenetic inhibition of HA neurons did notably reduce the ability of the wake-promoting compound modafinil to sustain wakefulness. Thus, part of modafinil's wake-promoting actions arise through the HA system.
Keywords: NREM sleep; caspase; chemogenetics; histamine; histidine decarboxylase; lesioning; modafinil; tuberomammillary nucleus; wakefulness.
© Sleep Research Society 2019. Published by Oxford University Press [on behalf of the Sleep Research Society].
Figures



References
-
- Lin JS. Brain structures and mechanisms involved in the control of cortical activation and wakefulness, with emphasis on the posterior hypothalamus and histaminergic neurons. Sleep Med Rev. 2000;4(5):471–503. - PubMed
-
- Haas H, et al. The role of histamine and the tuberomamillary nucleus in the nervous system. Nat Rev Neurosci. 2003;4(2):121–130. - PubMed
-
- Panula P, et al. The histaminergic network in the brain: basic organization and role in disease. Nat Rev Neurosci. 2013;14(7):472–487. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

