The ability to utilise ammonia as nitrogen source is cell type specific and intricately linked to GDH, AMPK and mTORC1
- PMID: 30728400
- PMCID: PMC6365639
- DOI: 10.1038/s41598-018-37509-3
The ability to utilise ammonia as nitrogen source is cell type specific and intricately linked to GDH, AMPK and mTORC1
Abstract
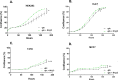
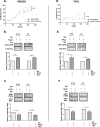
Ammonia can be utilised as an alternative nitrogen source to glutamine to support cell proliferation. However, the underlying molecular mechanisms and whether all cells have this ability is not fully understood. We find that eleven cancer and non-cancerous cell lines have opposite abilities to tolerate and utilise ammonia to support proliferation in a glutamine-depleted environment. HEK293, Huh7, T47D and MCF7 cells can use ammonia, when starved of glutamine, to support proliferation to varying degrees. Glutamine depletion reduced mTORC1 activity, while additional ammonia supplementation diminished this mTORC1 inhibition. Depletion of glutamine promoted a rapid and transient activation of AMPK, whereas, additional ammonia supplementation blocked this starvation-induced AMPK activation. As expected, drug-induced AMPK activation reduced cell proliferation in glutamine-depleted cells supplemented with ammonia. Surprisingly, mTORC1 activity was largely unchanged despite the enhanced AMPK activity, suggesting that AMPK does not inhibit mTORC1 signalling under these conditions. Finally, glutamate dehydrogenase (GDH) inhibition, a key enzyme regulating ammonia assimilation, leads to AMPK activation, mTORC1 inhibition and reduced proliferation. Ammonia provides an alternative nitrogen source that aids certain cancer cells ability to thrive in nutrient-deprived environment. The ability of cells to utilise ammonia as a nitrogen source is intricately linked to AMPK, mTORC1 and GDH.
Conflict of interest statement
The authors declare no competing interests.
Figures







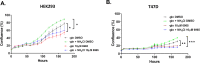
References
-
- Weisman, R. Target of Rapamycin (TOR) Regulates Growth in Response to Nutritional Signals. Microbiol Spectr4 (2016). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

