Solution Conformation of Bovine Leukemia Virus Gag Suggests an Elongated Structure
- PMID: 30731090
- PMCID: PMC6424597
- DOI: 10.1016/j.jmb.2019.01.036
Solution Conformation of Bovine Leukemia Virus Gag Suggests an Elongated Structure
Abstract
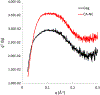
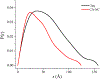
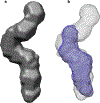
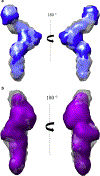
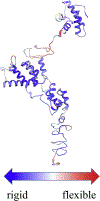
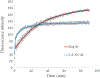
Bovine leukemia virus (BLV) is a deltaretrovirus that infects domestic cattle. The structural protein Gag, found in all retroviruses, is a polyprotein comprising three major functional domains: matrix (MA), capsid (CA), and nucleocapsid (NC). Previous studies have shown that both mature BLV MA and NC are able to bind to nucleic acids; however, the viral assembly process and packaging of viral genomic RNA requires full-length Gag to produce infectious particles. Compared to lentiviruses, little is known about the structure of the Gag polyprotein of deltaretroviruses. In this work, structural models of full-length BLV Gag and Gag lacking the MA domain were generated based on previous structural data of individual domains, homology modeling, and flexible fitting to SAXS data using molecular dynamics. The models were used in molecular dynamic simulations to determine the relative mobility of the protein backbone. Functional annealing assays revealed the role of MA in the nucleic acid chaperone activity of BLV Gag. Our results show that full-length BLV Gag has an elongated rod-shaped structure that is relatively rigid, with the exception of the linker between the MA and CA domains. Deletion of the MA domain maintains the elongated structure but alters the rate of BLV Gag-facilitated annealing of two complementary nucleic acids. These data are consistent with a role for the MA domain of retroviral Gag proteins in modulating nucleic acid binding and chaperone activity. IMPORTANCE: BLV is a retrovirus that is found worldwide in domestic cattle. Since BLV infection has serious implications for agriculture, and given its similarities to human retroviruses such as HTLV-1, the development of an effective treatment would have numerous benefits. The Gag polyprotein exists in all retroviruses and is a key player in viral assembly. However, the full-length structure of Gag from any virus has yet to be elucidated at high resolution. This study provides structural data for BLV Gag and could be a starting point for modeling Gag-small molecule interactions with the ultimate goal of developing of a new class of pharmaceuticals.
Keywords: Gag; matrix; molecular dynamics; retrovirus; small-angle X-ray scattering; viral assembly.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Figures

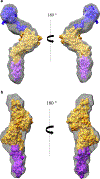
References
-
- Coffin JM, Hughes SH, Varmus HE. Retroviruses. Cold Spring Harbor (NY)1997. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

