Serum and Serum Albumin Inhibit in vitro Formation of Neutrophil Extracellular Traps (NETs)
- PMID: 30733715
- PMCID: PMC6354573
- DOI: 10.3389/fimmu.2019.00012
Serum and Serum Albumin Inhibit in vitro Formation of Neutrophil Extracellular Traps (NETs)
Abstract
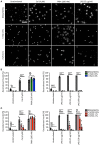
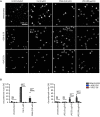
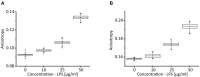
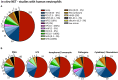
The formation of neutrophil extracellular traps (NETs) is an immune defense mechanism of neutrophilic granulocytes. Moreover, it is also involved in the pathogenesis of autoimmune, inflammatory, and neoplastic diseases. For that reason, the process of NET formation (NETosis) is subject of intense ongoing research. In vitro approaches to quantify NET formation are commonly used and involve neutrophil stimulation with various activators such as phorbol 12-myristate 13-acetate (PMA), lipopolysaccharides (LPS), or calcium ionophores (CaI). However, the experimental conditions of these experiments, particularly the media and media supplements employed by different research groups, vary considerably, rendering comparisons of results difficult. Here, we present the first standardized investigation of the influence of different media supplements on NET formation in vitro. The addition of heat-inactivated (hi) fetal calf serum (FCS), 0.5% human serum albumin (HSA), or 0.5% bovine serum albumin (BSA) efficiently prevented NET formation of human neutrophils following stimulation with LPS and CaI, but not after stimulation with PMA. Thus, serum components such as HSA, BSA and hiFCS (at concentrations typically found in the literature) inhibit NET formation to different degrees, depending on the NETosis inducer used. In contrast, in murine neutrophils, NETosis was inhibited by FCS and BSA, regardless of the inducer employed. This shows that mouse and human neutrophils have different susceptibilities toward the inhibition of NETosis by albumin or serum components. Furthermore, we provide experimental evidence that albumin inhibits NETosis by scavenging activators such as LPS. We also put our results into the context of media supplements most commonly used in NET research. In experiments with human neutrophils, either FCS (0.5-10%), heat-inactivated (hiFCS, 0.1-10%) or human serum albumin (HSA, 0.05-2%) was commonly added to the medium. For murine neutrophils, serum-free medium was used in most cases for stimulation with LPS and CaI, reflecting the different sensitivities of human and murine neutrophils to media supplements. Thus, the choice of media supplements greatly determines the outcome of experiments on NET-formation, which must be taken into account in NETosis research.
Keywords: NET; NETosis; experimental conditions; in vitro experiments; media; neutrophil extracelluar traps; neutrophils.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

