Evaluating CAR-T Cell Therapy in a Hypoxic 3D Tumor Model
- PMID: 30734529
- PMCID: PMC6448565
- DOI: 10.1002/adhm.201900001
Evaluating CAR-T Cell Therapy in a Hypoxic 3D Tumor Model
Abstract
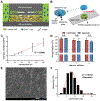
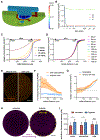
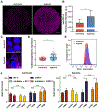
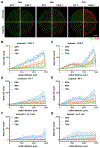
Despite its revolutionary success in hematological malignancies, chimeric antigen receptor T (CAR-T) cell therapy faces disappointing clinical results in solid tumors. The poor efficacy has been partially attributed to the lack of understanding in how CAR-T cells function in a solid tumor microenvironment. Hypoxia plays a critical role in cancer progression and immune editing, which potentially results in solid tumors escaping immunosurveillance and CAR-T cell-mediated cytotoxicity. Mechanistic studies of CAR-T cell biology in a physiological environment has been limited by the complexity of tumor-immune interactions in clinical and animal models, as well as by a lack of reliable in vitro models. A microdevice platform that recapitulates a 3D tumor section with a gradient of oxygen and integrates fluidic channels surrounding the tumor for CAR-T cell delivery is engineered. The design allows for the evaluation of CAR-T cell cytotoxicity and infiltration in the heterogeneous oxygen landscape of in vivo solid tumors at a previously unachievable scale in vitro.
Keywords: chimeric antigen receptors; hypoxia; immune checkpoints; immunotherapy; ovarian cancer; solid tumors.
© 2019 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
Conflict of Interest
K.S., Y.A., and H.P.T. are listed as inventors for a US patent application disclosing the hypoxia device, which includes part of the data described in the manuscript.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

