Improving the Production of Salt-Tolerant Glutaminase by Integrating Multiple Copies of Mglu into the Protease and 16S rDNA Genes of Bacillus subtilis 168
- PMID: 30736411
- PMCID: PMC6384544
- DOI: 10.3390/molecules24030592
Improving the Production of Salt-Tolerant Glutaminase by Integrating Multiple Copies of Mglu into the Protease and 16S rDNA Genes of Bacillus subtilis 168
Abstract
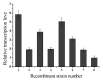
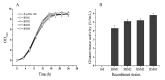
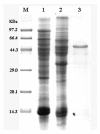
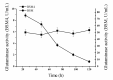
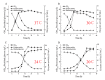
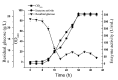
In this study, the Micrococcus luteus K-3 glutaminase was successfully over-expressed in the GRAS (Generally Recognized as Safe) Bacillus subtilis strain 168 by integration of the Mglu gene in the 16S rDNA locus. This was done in order to screen a strain producing high levels of recombinant glutaminase from the selected candidates. The transcription of the glutaminase genes in the B. subtilis 168 chromosome and the expression of glutaminase protein was further assessed by qPCR, SDS-PAGE analysis and an enzyme activity assay. To further increase the production of glutaminase, the nprB and nprE genes, which encode specific proteases, were disrupted by integration of the Mglu gene. After continuous cell culturing without the addition of antibiotics, the integrated recombinant strains showed excellent genetic stability, demonstrating favorable industrialization potential. After the fermentation temperature was optimized, a 5-L bioreactor was used for fed-batch fermentation of the recombinant glutaminase producing strain at 24 °C, and the highest enzyme activity achieved was approximately 357.6 U/mL.
Keywords: 16S rDNA; Bacillus subtilis; glutaminase; integrated expression.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

References
-
- Wakayama M., Nagano Y., Renu N., Kawamura T., Sakai K., Moriguchi M. Molecular cloning and determination of the nucleotide sequence of a gene encoding salt-tolerant glutaminase from Micrococcus luteus K-3. J. Ferment. Bioeng. 1996;82:592–597. doi: 10.1016/S0922-338X(97)81259-7. - DOI
MeSH terms
Substances
Grants and funding
- 31500065, 31570085/National Natural Science Foundation of China
- JUSRP51708A/Fundamental Research Funds for the Central Universities
- KLIB-KF201703/the Key Laboratory of Industrial Biotechnology, Ministry of Education, China
- SKLF-KF-201709/the Open Project Program of State Key Laboratory of Food Science and Technology, Jiangnan University
- 111-2-06/the Priority Academic Program Development of Jiangsu Higher Education Institutions, the 111 Project
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

