Drug susceptibility testing and mortality in patients treated for tuberculosis in high-burden countries: a multicentre cohort study
- PMID: 30744962
- PMCID: PMC6482382
- DOI: 10.1016/S1473-3099(18)30673-X
Drug susceptibility testing and mortality in patients treated for tuberculosis in high-burden countries: a multicentre cohort study
Abstract
Background: Drug resistance is a challenge for the global control of tuberculosis. We examined mortality in patients with tuberculosis from high-burden countries, according to concordance or discordance of results from drug susceptibility testing done locally and in a reference laboratory.
Methods: This multicentre cohort study was done in Côte d'Ivoire, Democratic Republic of the Congo, Kenya, Nigeria, South Africa, Peru, and Thailand. We collected Mycobacterium tuberculosis isolates and clinical data from adult patients aged 16 years or older. Patients were stratified by HIV status and tuberculosis drug resistance. Molecular or phenotypic drug susceptibility testing was done locally and at the Swiss National Center for Mycobacteria, Zurich, Switzerland. We examined mortality during treatment according to drug susceptibility test results and treatment adequacy in multivariable logistic regression models adjusting for sex, age, sputum microscopy, and HIV status.
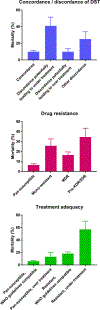
Findings: We obtained M tuberculosis isolates from 871 patients diagnosed between 2013 and 2016. After exclusion of 237 patients, 634 patients with tuberculosis were included in this analysis; the median age was 33·2 years (IQR 26·9-42·5), 239 (38%) were women, 272 (43%) were HIV-positive, and 69 (11%) patients died. Based on the reference laboratory drug susceptibility test, 394 (62%) strains were pan-susceptible, 45 (7%) monoresistant, 163 (26%) multidrug-resistant (MDR), and 30 (5%) had pre-extensively or extensively drug resistant (pre-XDR or XDR) tuberculosis. Results of reference and local laboratories were concordant for 513 (81%) of 634 patients and discordant for 121 (19%) of 634. Overall, sensitivity to detect any resistance was 90·8% (95% CI 86·5-94·2) and specificity 84·3% (80·3-87·7). Mortality ranged from 6% (20 of 336) in patients with pan-susceptible tuberculosis treated according to WHO guidelines to 57% (eight of 14) in patients with resistant strains who were under-treated. In logistic regression models, compared with concordant drug susceptibility test results, the adjusted odds ratio of death was 7·33 (95% CI 2·70-19·95) for patients with discordant results potentially leading to under-treatment.
Interpretation: Inaccurate drug susceptibility testing by comparison with a reference standard leads to under-treatment of drug-resistant tuberculosis and increased mortality. Rapid molecular drug susceptibility test of first-line and second-line drugs at diagnosis is required to improve outcomes in patients with MDR tuberculosis and pre-XDR or XDR tuberculosis.
Funding: National Institutes of Allergy and Infectious Diseases, Swiss National Science Foundation, Swiss National Center for Mycobacteria.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Conflict of interest statement
CONFLICTS OF INTEREST
AA has received honoraria fees from Jensen-Cilag, Gilead and Bristol-Myers Squibb. All other authors have no conflicts of interest to declare.
Figures
Comment in
-
Universal regimens or universal access to drug susceptibility testing for tuberculosis?Lancet Infect Dis. 2019 Mar;19(3):224-225. doi: 10.1016/S1473-3099(18)30742-4. Epub 2019 Feb 8. Lancet Infect Dis. 2019. PMID: 30744961 No abstract available.
References
-
- World Health Organization. Global Tuberculosis Report 2017. Geneva, 2017. DOI:WHO/HTM/TB/2017.23.
-
- Antiretroviral Therapy in Low-Income Countries Collaboration of the International epidemiological Databases to Evaluate AIDS (IeDEA),ART Cohort Collaboration, Brinkhof MWG, et al. Tuberculosis after Initiation of Antiretroviral Therapy in Low-Income and High-Income Countries. Clin Infect Dis 2007; 45: 1518–21. - PMC - PubMed
-
- Lawn SD, Wood R, De Cock KM, Kranzer K, Lewis JJ, Churchyard GJ. Antiretrovirals and isoniazid preventive therapy in the prevention of HIV-associated tuberculosis in settings with limited health-care resources. Lancet Infect Dis 2010; 10: 489–98. - PubMed

