Focus on Cdc42 in Breast Cancer: New Insights, Target Therapy Development and Non-Coding RNAs
- PMID: 30754684
- PMCID: PMC6406589
- DOI: 10.3390/cells8020146
Focus on Cdc42 in Breast Cancer: New Insights, Target Therapy Development and Non-Coding RNAs
Abstract
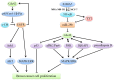
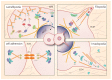
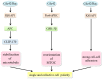
Breast cancer is the most common malignant tumors in females. Although the conventional treatment has demonstrated a certain effect, some limitations still exist. The Rho guanosine triphosphatase (GTPase) Cdc42 (Cell division control protein 42 homolog) is often upregulated by some cell surface receptors and oncogenes in breast cancer. Cdc42 switches from inactive guanosine diphosphate (GDP)-bound to active GTP-bound though guanine-nucleotide-exchange factors (GEFs), results in activation of signaling cascades that regulate various cellular processes such as cytoskeletal changes, proliferation and polarity establishment. Targeting Cdc42 also provides a strategy for precise breast cancer therapy. In addition, Cdc42 is a potential target for several types of non-coding RNAs including microRNAs and lncRNAs. These non-coding RNAs is extensively involved in Cdc42-induced tumor processes, while many of them are aberrantly expressed. Here, we focus on the role of Cdc42 in cell morphogenesis, proliferation, motility, angiogenesis and survival, introduce the Cdc42-targeted non-coding RNAs, as well as present current development of effective Cdc42-targeted inhibitors in breast cancer.
Keywords: Cdc42; breast cancer; cytoskeleton remodeling; non-coding RNAs; targeted therapy; tumor progression.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

