Semaphorin 3C as a Therapeutic Target in Prostate and Other Cancers
- PMID: 30759745
- PMCID: PMC6386986
- DOI: 10.3390/ijms20030774
Semaphorin 3C as a Therapeutic Target in Prostate and Other Cancers
Abstract
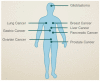
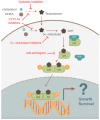
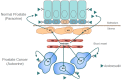
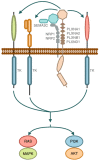
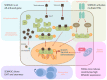
The semaphorins represent a large family of signaling molecules with crucial roles in neuronal and cardiac development. While normal semaphorin function pertains largely to development, their involvement in malignancy is becoming increasingly evident. One member, Semaphorin 3C (SEMA3C), has been shown to drive a number of oncogenic programs, correlate inversely with cancer prognosis, and promote the progression of multiple different cancer types. This report surveys the body of knowledge surrounding SEMA3C as a therapeutic target in cancer. In particular, we summarize SEMA3C's role as an autocrine andromedin in prostate cancer growth and survival and provide an overview of other cancer types that SEMA3C has been implicated in including pancreas, brain, breast, and stomach. We also propose molecular strategies that could potentially be deployed against SEMA3C as anticancer agents such as biologics, small molecules, monoclonal antibodies and antisense oligonucleotides. Finally, we discuss important considerations for the inhibition of SEMA3C as a cancer therapeutic agent.
Keywords: SEMA3C; cancer therapeutics; inhibitors; neuropilins; plexins; semaphorins.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

