Peripheral Venous Blood Platelet-to-Lymphocyte Ratio (PLR) for Predicting the Survival of Patients With Gastric Cancer Treated With SOX or XELOX Regimen Neoadjuvant Chemotherapy
- PMID: 30760114
- PMCID: PMC6378642
- DOI: 10.1177/1533033819829485
Peripheral Venous Blood Platelet-to-Lymphocyte Ratio (PLR) for Predicting the Survival of Patients With Gastric Cancer Treated With SOX or XELOX Regimen Neoadjuvant Chemotherapy
Abstract
Background: Inflammation plays an important role in tumor progression. Predicting survival is remarkably difficult in patients with gastric cancer receiving neoadjuvant chemotherapy. The aim of the present study is to investigate the potential prognostic significance of the platelet-to-lymphocyte ratio in patients with gastric cancer receiving S-1 plus oxaliplatin or oxaliplatin and capecitabine regimen.
Methods: Ninety-one patients with gastric cancer treated with neoadjuvant chemotherapy were enrolled in this study and then underwent operation. The optimal cutoff value was calculated using receiver-operating characteristic curve analyses. The optimal cutoff value of platelet-to-lymphocyte ratio was divided into low platelet-to-lymphocyte ratio <162 group and high platelet-to-lymphocyte ratio ≥162 group. Kaplan-Meier method and log-rank test were used to analyze the survival curves. The independent prognostic factors and prognostic value of the platelet-to-lymphocyte ratio were assessed by univariate and multivariate Cox proportional hazards regression model. The toxicity was evaluated according to the National Cancer Institute Common Toxicity Criteria.
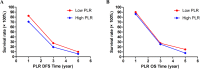
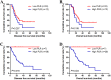
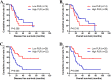
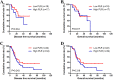
Results: Kaplan-Meier analyses revealed that patients with low platelet-to-lymphocyte ratio correlated remarkably with better mean disease-free survival and mean overall survival than those with high platelet-to-lymphocyte ratio (mean disease-free survival 47.33 and 33.62 months, respectively; mean overall survival 51.21 and 36.80 months, respectively). The results demonstrated that platelet-to-lymphocyte ratio had prognostic significance using the cutoff value of 162 on disease-free survival and overall survival, and the mean disease-free survival and overall survival time for patients with low platelet-to-lymphocyte ratio were longer than those with high platelet-to-lymphocyte ratio. Meanwhile, patients with gastric cancer who had lower platelet-to-lymphocyte ratio had longer 1-, 3-, and 5-year rates of disease-free survival and overall survival. Moreover, patients with low platelet-to-lymphocyte ratio had longer mean disease-free survival and overall survival than those with high platelet-to-lymphocyte ratio in receiving S-1 plus oxaliplatin or oxaliplatin and capecitabine regimen.
Conclusions: The preoperative platelet-to-lymphocyte ratio may be a promising and convenient prognostic biomarker for patients gastric cancer receiving S-1 plus oxaliplatin or oxaliplatin and capecitabine regimen neoadjuvant chemotherapy. It may be useful to help the doctors identify the high-risk patients for taking efficient treatment strategy decisions.
Keywords: S-1 plus oxaliplatin; gastric cancer; neoadjuvant chemotherapy; oxaliplatin and capecitabine; platelet-to-lymphocyte ratio.
Conflict of interest statement
Figures

Similar articles
-
[Safety and effectiveness of oxaliplatin combined with capecitabine or oxaliplatin combined with S-1 neoadjuvant chemotherapy in the treatment of advanced gastric cancer].Zhonghua Wei Chang Wai Ke Za Zhi. 2021 Feb 25;24(2):138-144. doi: 10.3760/cma.j.cn.441530-20200721-00433. Zhonghua Wei Chang Wai Ke Za Zhi. 2021. PMID: 33508919 Chinese.
-
[Safety and efficacy of adjuvant chemotherapy with oxaliplatin and S-1 for patients with locally advanced gastric cancer after D2 lymph nodes dissection].Zhonghua Wei Chang Wai Ke Za Zhi. 2021 Feb 25;24(2):145-152. doi: 10.3760/cma.j.cn.441530-20201016-00561. Zhonghua Wei Chang Wai Ke Za Zhi. 2021. PMID: 33508920 Chinese.
-
In Gastric Cancer Patients Receiving Neoadjuvant Chemotherapy Systemic Inflammation Response Index is a Useful Prognostic Indicator.Pathol Oncol Res. 2021 Oct 12;27:1609811. doi: 10.3389/pore.2021.1609811. eCollection 2021. Pathol Oncol Res. 2021. PMID: 34712105 Free PMC article.
-
[Progress in chemotherapy for advanced gastric cancer].Ai Zheng. 2009 Oct;28(10):1108-13. doi: 10.5732/cjc.008.10560. Ai Zheng. 2009. PMID: 19799823 Review. Chinese.
-
Evaluation of the prognostic role of platelet-lymphocyte ratio in cancer patients treated with immune checkpoint inhibitors: A systematic review and meta-analysis.Int Immunopharmacol. 2019 Dec;77:105957. doi: 10.1016/j.intimp.2019.105957. Epub 2019 Oct 31. Int Immunopharmacol. 2019. PMID: 31677498
Cited by
-
The Value of the C-Reactive Protein-to-Lymphocyte Ratio for Predicting Lymphovascular Invasion Based on Nutritional Status in Gastric Cancer.Technol Cancer Res Treat. 2022 Jan-Dec;21:15330338221106517. doi: 10.1177/15330338221106517. Technol Cancer Res Treat. 2022. PMID: 35695253 Free PMC article.
-
A Nomogram Based on Hematological Parameters and Clinicopathological Characteristics for Predicting Local-Regional Recurrence After Breast-Conserving Therapy.Front Oncol. 2022 Jul 19;12:861210. doi: 10.3389/fonc.2022.861210. eCollection 2022. Front Oncol. 2022. PMID: 35928880 Free PMC article.
-
Analysis of the safety and efficacy of laparoscopic gastrojejunostomy following neoadjuvant chemotherapy for gastric pyloric obstruction.Front Oncol. 2025 Mar 18;15:1430761. doi: 10.3389/fonc.2025.1430761. eCollection 2025. Front Oncol. 2025. PMID: 40171263 Free PMC article.
-
Prognostic Value of Neutrophil-Lymphocyte Ratio, Platelet-Lymphocyte Ratio, and Combined Neutrophil-Lymphocyte Ratio and Platelet-Lymphocyte Ratio in Stage IV Advanced Gastric Cancer.Front Oncol. 2020 Jun 19;10:841. doi: 10.3389/fonc.2020.00841. eCollection 2020. Front Oncol. 2020. PMID: 32637353 Free PMC article.
-
The Clinical Impact of the Pretreatment Platelet-to-Lymphocyte Ratio in Gastric Cancer Patients Who Receive Curative Treatment.In Vivo. 2023 Nov-Dec;37(6):2696-2703. doi: 10.21873/invivo.13379. In Vivo. 2023. PMID: 37905661 Free PMC article.
References
-
- Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. - PubMed
-
- Shen L, Shan YS, Hu HM, et al. Management of gastric cancer in Asia: resource-stratified guidelines. Lancet Oncol. 2013;14(12):e535–e547. - PubMed
-
- Pera M, Gallego R, Montagut C, et al. Phase II trial of preoperative chemoradiotherapy with oxaliplatin, cisplatin, and 5-FU in locally advanced esophageal and gastric cancer. Ann Oncol. 2012;23(3):664–670. - PubMed
-
- Wang X, Wang ML, Zhou LY, Lu XY, Yang JF, Yu HG. Randomized phase II study comparing paclitaxel with S-1 vs. S-1 as first-line treatment in patients with advanced gastric cancer. Clin Transl Oncol. 2013;15(10):836–842. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

