TMEM16A in Cystic Fibrosis: Activating or Inhibiting?
- PMID: 30761000
- PMCID: PMC6362895
- DOI: 10.3389/fphar.2019.00003
TMEM16A in Cystic Fibrosis: Activating or Inhibiting?
Abstract
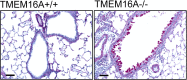
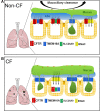
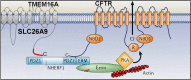
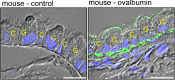
The inflammatory airway disease cystic fibrosis (CF) is characterized by airway obstruction due to mucus hypersecretion, airway plugging, and bronchoconstriction. The cystic fibrosis transmembrane conductance regulator (CFTR) chloride channel is dysfunctional in CF, leading to defects in epithelial transport. Although CF pathogenesis is still disputed, activation of alternative Cl- channels is assumed to improve lung function in CF. Two suitable non-CFTR Cl- channels are present in the airway epithelium, the Ca2+ activated channel TMEM16A and SLC26A9. Activation of these channels is thought to be feasible to improve hydration of the airway mucus and to increase mucociliary clearance. Interestingly, both channels are upregulated during inflammatory lung disease. They are assumed to support fluid secretion, necessary to hydrate excess mucus and to maintain mucus clearance. During inflammation, however, TMEM16A is upregulated particularly in mucus producing cells, with only little expression in ciliated cells. Recently it was shown that knockout of TMEM16A in ciliated cells strongly compromises Cl- conductance and attenuated mucus secretion, but does not lead to a CF-like lung disease and airway plugging. Along this line, activation of TMEM16A by denufosol, a stable purinergic ligand, failed to demonstrate any benefit to CF patients in earlier studies. It rather induced adverse effects such as cough. A number of studies suggest that TMEM16A is essential for mucus secretion and possibly also for mucus production. Evidence is now provided for a crucial role of TMEM16A in fusion of mucus-filled granules with the apical plasma membrane and cellular exocytosis. This is probably due to local Ca2+ signals facilitated by TMEM16A. Taken together, TMEM16A supports fluid secretion by ciliated airway epithelial cells, but also maintains excessive mucus secretion during inflammatory airway disease. Because TMEM16A also supports airway smooth muscle contraction, inhibition rather than activation of TMEM16A might be the appropriate treatment for CF lung disease, asthma and COPD. As a number of FDA-approved and well-tolerated drugs have been shown to inhibit TMEM16A, evaluation in clinical trials appears timely.
Keywords: COPD; Ca2+ signaling; TMEM16A; anoctamin 1; asthma; cystic fibrosis; mucus secretion.
Figures


Similar articles
-
TMEM16A Mediates Mucus Production in Human Airway Epithelial Cells.Am J Respir Cell Mol Biol. 2021 Jan;64(1):50-58. doi: 10.1165/rcmb.2019-0442OC. Am J Respir Cell Mol Biol. 2021. PMID: 33026825
-
Mucus Release and Airway Constriction by TMEM16A May Worsen Pathology in Inflammatory Lung Disease.Int J Mol Sci. 2021 Jul 22;22(15):7852. doi: 10.3390/ijms22157852. Int J Mol Sci. 2021. PMID: 34360618 Free PMC article.
-
TMEM16A Potentiation: A Novel Therapeutic Approach for the Treatment of Cystic Fibrosis.Am J Respir Crit Care Med. 2020 Apr 15;201(8):946-954. doi: 10.1164/rccm.201908-1641OC. Am J Respir Crit Care Med. 2020. PMID: 31898911 Free PMC article.
-
Pharmacotherapy of the ion transport defect in cystic fibrosis: role of purinergic receptor agonists and other potential therapeutics.Am J Respir Med. 2003;2(4):299-309. doi: 10.1007/BF03256658. Am J Respir Med. 2003. PMID: 14719996 Review.
-
TMEM16A (ANO1) as a therapeutic target in cystic fibrosis.Curr Opin Pharmacol. 2022 Jun;64:102206. doi: 10.1016/j.coph.2022.102206. Epub 2022 Mar 29. Curr Opin Pharmacol. 2022. PMID: 35364521 Review.
Cited by
-
Paneth Cell Secretion in vivo Requires Expression of Tmem16a and Tmem16f.Gastro Hep Adv. 2022 Aug 7;1(6):1088-1098. doi: 10.1016/j.gastha.2022.08.002. eCollection 2022. Gastro Hep Adv. 2022. PMID: 39131261 Free PMC article.
-
Chloride transport modulators as drug candidates.Am J Physiol Cell Physiol. 2021 Dec 1;321(6):C932-C946. doi: 10.1152/ajpcell.00334.2021. Epub 2021 Oct 13. Am J Physiol Cell Physiol. 2021. PMID: 34644122 Free PMC article. Review.
-
TMEM16A: An Alternative Approach to Restoring Airway Anion Secretion in Cystic Fibrosis?Int J Mol Sci. 2020 Mar 30;21(7):2386. doi: 10.3390/ijms21072386. Int J Mol Sci. 2020. PMID: 32235608 Free PMC article. Review.
-
Development of a QPatch-Automated Electrophysiology Assay for Identifying TMEM16A Small-Molecule Inhibitors.Assay Drug Dev Technol. 2020 Apr;18(3):134-147. doi: 10.1089/adt.2019.962. Assay Drug Dev Technol. 2020. PMID: 32319819 Free PMC article.
-
TMEM16A drives renal cyst growth by augmenting Ca2+ signaling in M1 cells.J Mol Med (Berl). 2020 May;98(5):659-671. doi: 10.1007/s00109-020-01894-y. Epub 2020 Mar 18. J Mol Med (Berl). 2020. PMID: 32185407 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

