Severe Acute Respiratory Syndrome Coronavirus Viroporin 3a Activates the NLRP3 Inflammasome
- PMID: 30761102
- PMCID: PMC6361828
- DOI: 10.3389/fmicb.2019.00050
Severe Acute Respiratory Syndrome Coronavirus Viroporin 3a Activates the NLRP3 Inflammasome
Abstract
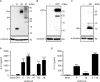
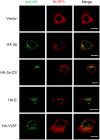
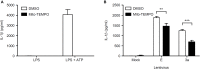
Nod-like receptor family, pyrin domain-containing 3 (NLRP3) regulates the secretion of proinflammatory cytokines interleukin 1 beta (IL-1β) and IL-18. We previously showed that influenza virus M2 or encephalomyocarditis virus (EMCV) 2B proteins stimulate IL-1β secretion following activation of the NLRP3 inflammasome. However, the mechanism by which severe acute respiratory syndrome coronavirus (SARS-CoV) activates the NLRP3 inflammasome remains unknown. Here, we provide direct evidence that SARS-CoV 3a protein activates the NLRP3 inflammasome in lipopolysaccharide-primed macrophages. SARS-CoV 3a was sufficient to cause the NLRP3 inflammasome activation. The ion channel activity of the 3a protein was essential for 3a-mediated IL-1β secretion. While cells uninfected or infected with a lentivirus expressing a 3a protein defective in ion channel activity expressed NLRP3 uniformly throughout the cytoplasm, NLRP3 was redistributed to the perinuclear space in cells infected with a lentivirus expressing the 3a protein. K+ efflux and mitochondrial reactive oxygen species were important for SARS-CoV 3a-induced NLRP3 inflammasome activation. These results highlight the importance of viroporins, transmembrane pore-forming viral proteins, in virus-induced NLRP3 inflammasome activation.
Keywords: IL-1β; SARS-CoV; inflammasome; inflammation; viroporin.
Figures



References
-
- Castano-Rodriguez C., Honrubia J. M., Gutierrez-Alvarez J., Dediego M. L., Nieto-Torres J. L., Jimenez-Guardeno J. M., et al. (2018). Role of severe acute respiratory syndrome Coronavirus viroporins E, 3a, and 8a in replication and pathogenesis. mBio 9:e2325–17. 10.1128/mBio.02325-17 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

