Coenzyme Q10 protects against burn-induced mitochondrial dysfunction and impaired insulin signaling in mouse skeletal muscle
- PMID: 30761259
- PMCID: PMC6356165
- DOI: 10.1002/2211-5463.12580
Coenzyme Q10 protects against burn-induced mitochondrial dysfunction and impaired insulin signaling in mouse skeletal muscle
Abstract
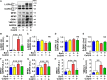
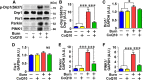
Mitochondrial dysfunction is associated with metabolic alterations in various disease states, including major trauma (e.g., burn injury). Metabolic derangements, including muscle insulin resistance and hyperlactatemia, are a clinically significant complication of major trauma. Coenzyme Q10 (CoQ10) is an essential cofactor for mitochondrial electron transport, and its reduced form acts as a lipophilic antioxidant. Here, we report that burn injury induces impaired muscle insulin signaling, hyperlactatemia, mitochondrial dysfunction (as indicated by suppressed mitochondrial oxygen consumption rates), morphological alterations of the mitochondria (e. g., enlargement, and loss of cristae structure), mitochondrial oxidative stress, and disruption of mitochondrial integrity (as reflected by increased mitochondrial DNA levels in the cytosol and circulation). All of these alterations were significantly alleviated by CoQ10 treatment compared with vehicle alone. These findings indicate that CoQ10 treatment is efficacious in protecting against mitochondrial dysfunction and insulin resistance in skeletal muscle of burned mice. Our data highlight CoQ10 as a potential new strategy to prevent mitochondrial damage and metabolic dysfunction in burn patients.
Keywords: burn injury; coenzyme Q10; insulin resistance; mitochondrial dysfunction; skeletal muscle.
Figures




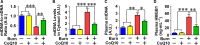



Similar articles
-
Role of protein farnesylation in burn-induced metabolic derangements and insulin resistance in mouse skeletal muscle.PLoS One. 2015 Jan 16;10(1):e0116633. doi: 10.1371/journal.pone.0116633. eCollection 2015. PLoS One. 2015. PMID: 25594415 Free PMC article.
-
Burn-induced muscle metabolic derangements and mitochondrial dysfunction are associated with activation of HIF-1α and mTORC1: Role of protein farnesylation.Sci Rep. 2017 Jul 26;7(1):6618. doi: 10.1038/s41598-017-07011-3. Sci Rep. 2017. PMID: 28747716 Free PMC article.
-
Inhibition of oxidative stress by coenzyme Q10 increases mitochondrial mass and improves bioenergetic function in optic nerve head astrocytes.Cell Death Dis. 2013 Oct 3;4(10):e820. doi: 10.1038/cddis.2013.341. Cell Death Dis. 2013. PMID: 24091663 Free PMC article.
-
[Coenzyme Q10: its biosynthesis and biological significance in animal organisms and in humans].Postepy Hig Med Dosw (Online). 2005;59:150-9. Postepy Hig Med Dosw (Online). 2005. PMID: 15928598 Review. Polish.
-
Coenzyme Q10 treatment of cardiovascular disorders of ageing including heart failure, hypertension and endothelial dysfunction.Clin Chim Acta. 2015 Oct 23;450:83-9. doi: 10.1016/j.cca.2015.08.002. Epub 2015 Aug 6. Clin Chim Acta. 2015. PMID: 26254995 Review.
Cited by
-
The role of mtDAMPs in the trauma-induced systemic inflammatory response syndrome.Front Immunol. 2023 Jul 18;14:1164187. doi: 10.3389/fimmu.2023.1164187. eCollection 2023. Front Immunol. 2023. PMID: 37533869 Free PMC article. Review.
-
Farnesysltransferase Inhibitor Prevents Burn Injury-Induced Metabolome Changes in Muscle.Metabolites. 2022 Aug 27;12(9):800. doi: 10.3390/metabo12090800. Metabolites. 2022. PMID: 36144205 Free PMC article.
-
Efficient expansion and delayed senescence of hUC-MSCs by microcarrier-bioreactor system.Stem Cell Res Ther. 2023 Oct 4;14(1):284. doi: 10.1186/s13287-023-03514-1. Stem Cell Res Ther. 2023. PMID: 37794520 Free PMC article.
-
Mitochondrial Fission as a Therapeutic Target for Metabolic Diseases: Insights into Antioxidant Strategies.Antioxidants (Basel). 2023 May 27;12(6):1163. doi: 10.3390/antiox12061163. Antioxidants (Basel). 2023. PMID: 37371893 Free PMC article. Review.
-
Metabolomic and proteomic profiling of a burn-hemorrhagic shock swine model reveals a metabolomic signature associated with fatal outcomes.Eur J Med Res. 2025 Jan 7;30(1):10. doi: 10.1186/s40001-024-02245-0. Eur J Med Res. 2025. PMID: 39773520 Free PMC article.
References
-
- Cochran A, Edelman LS, Saffle JR and Morris SE (2007) The relationship of serum lactate and base deficit in burn patients to mortality. J Burn Care Res 28, 231–240. - PubMed
-
- Gore DC, Ferrando A, Barnett J, Wolf SE, Desai M, Herndon DN, Goodwin C & Wolfe RR (2000) Influence of glucose kinetics on plasma lactate concentration and energy expenditure in severely burned patients. J Trauma 49, 673–677; discussion 677‐8. - PubMed
-
- Brealey D, Brand M, Hargreaves I, Heales S, Land J, Smolenski R, Davies NA, Cooper CE and Singer M (2002) Association between mitochondrial dysfunction and severity and outcome of septic shock. Lancet 360, 219–223. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

