Neurobiology of Photophobia
- PMID: 30762717
- PMCID: PMC6383812
- DOI: 10.1097/WNO.0000000000000766
Neurobiology of Photophobia
Abstract
Background: Photophobia is commonly associated with migraine, meningitis, concussion, and a variety of ocular diseases. Advances in our ability to trace multiple brain pathways through which light information is processed have paved the way to a better understanding of the neurobiology of photophobia and the complexity of the symptoms triggered by light.
Purpose: The purpose of this review is to summarize recent anatomical and physiological studies on the neurobiology of photophobia with emphasis on migraine.
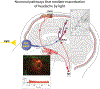
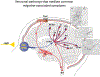
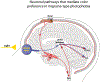
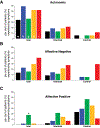
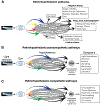
Recent findings: Observations made in blind and seeing migraine patients, and in a variety of animal models, have led to the discovery of a novel retino-thalamo-cortical pathway that carries photic signal from melanopsinergic and nonmelanopsinergic retinal ganglion cells (RGCs) to thalamic neurons. Activity of these neurons is driven by migraine and their axonal projections convey signals about headache and light to multiple cortical areas involved in the generation of common migraine symptoms. Novel projections of RGCs into previously unidentified hypothalamic neurons that regulate parasympathetic and sympathetic functions have also been discovered. Finally, recent work has led to a novel understanding of color preference in migraine-type photophobia and of the roles played by the retina, thalamus, and cortex.
Summary: The findings provide a neural substrate for understanding the complexity of aversion to light in patients with migraine and neuro-ophthalmologic other disorders.
Conflict of interest statement
Figures

References
-
- Lebensohn JE: Photophobia: mechanism and implications. Am J Ophthalmol 1951, 34:1294–1300. - PubMed
-
- Kawasaki A, Purvin VA: Photophobia as the presenting visual symptom of chiasmal compression. J Neuroophthalmol 2002, 22:3–8. - PubMed
-
- Aurora SK, Cao Y, Bowyer SM, Welch KM: The occipital cortex is hyperexcitable in migraine: experimental evidence. Headache 1999, 39:469–476. - PubMed
-
- Lamonte M, Silberstein SD, Marcelis JF: Headache associated with aseptic meningitis. Headache 1995, 35:520–526. - PubMed
-
- Welty TE, Horner TG: Pathophysiology and treatment of subarachnoid hemorrhage. Clin Pharm 1990, 9:35–39. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

