Particle-Attachment-Mediated and Matrix/Lattice-Guided Enamel Apatite Crystal Growth
- PMID: 30763075
- PMCID: PMC7067265
- DOI: 10.1021/acsnano.8b08668
Particle-Attachment-Mediated and Matrix/Lattice-Guided Enamel Apatite Crystal Growth
Abstract
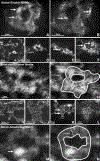
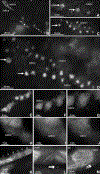
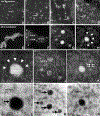
Tooth enamel is a hard yet resilient biomaterial that derives its unique mechanical properties from decussating bundles of apatite crystals. To understand enamel crystal nucleation and growth at a nanoscale level and to minimize preparation artifacts, the developing mouse enamel matrix was imaged in situ using graphene liquid cells and atomic resolution scanning transmission electron and cryo-fracture electron microscopy. We report that 1-2 nm diameter mineral precipitates aggregated to form larger 5 nm particle assemblies within ameloblast secretory vesicles or annular organic matrix subunits. Further evidence for the fusion of 1-2 nm mineral precipitates into 5 nm mineral aggregates via particle attachment was provided by matrix-mediated calcium phosphate crystal growth studies. As a next step, aggregated particles organized into rows of 3-10 subunits and developed lattice suprastructures with 0.34 nm gridline spacings corresponding to the (002) planes of apatite crystals. Mineral lattice suprastructures superseded closely matched organic matrix patterns, suggestive of a combination of organic/inorganic templates guiding apatite crystal growth. Upon assembly of 2-5 nm subunits into crystal ribbons, lattice fringes indicative of the presence of larger ordered crystallites were observed surrounding elongating crystal ribbons, presumably guiding the c-axis growth of composite apatite crystals. Cryo-fracture micrographs revealed reticular networks of an organic matrix on the surface of elongating enamel crystal ribbons, suggesting that protein coats facilitate c-axis apatite crystal growth. Together, these data demonstrate (i) the involvement of particle attachment in enamel crystal nucleation, (ii) a combination of matrix- and lattice-guided crystal growth, and (iii) fusion of individual crystals via a mechanism similar to Ostwald ripening.
Keywords: apatite; atomic scale microscopy; crystal growth; enamel; graphene liquid cell.
Figures



References
-
- Lowenstam HA Minerals Formed by Organisms. Science 1981, 211, 1126–1131. - PubMed
-
- Mann S Biomineralization: Principles and Concepts in Bioinorganic Materials Chemistry Oxford University Press: Oxford, UK, 2001; pp 3.
-
- Diekwisch T; David S; Bringas P Jr; Santos V; Slavkin HC Antisense Inhibition of AMEL Translation Demonstrates Supramolecular Controls for Enamel HAP Crystal Growth during Embryonic Mouse Molar Development. Development 1993, 117, 471–482. - PubMed
-
- Diekwisch TG; Berman BJ; Gentner S; Slavkin HC Initial Enamel Crystals are not Spatially Associated with Mineralized Dentine. Cell Tissue Res 1995, 279, 49–67. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

