Influence of Platelet-Rich and Platelet-Poor Plasma on Endogenous Mechanisms of Skeletal Muscle Repair/Regeneration
- PMID: 30764506
- PMCID: PMC6387315
- DOI: 10.3390/ijms20030683
Influence of Platelet-Rich and Platelet-Poor Plasma on Endogenous Mechanisms of Skeletal Muscle Repair/Regeneration
Abstract
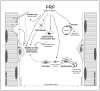
The morpho-functional recovery of injured skeletal muscle still represents an unmet need. None of the therapeutic options so far adopted have proved to be resolutive. A current scientific challenge remains the identification of effective strategies improving the endogenous skeletal muscle regenerative program. Indeed, skeletal muscle tissue possesses an intrinsic remarkable regenerative capacity in response to injury, mainly thanks to the activity of a population of resident muscle progenitors called satellite cells, largely influenced by the dynamic interplay established with different molecular and cellular components of the surrounding niche/microenvironment. Other myogenic non-satellite cells, residing within muscle or recruited via circulation may contribute to post-natal muscle regeneration. Unfortunately, in the case of extended damage the tissue repair may become aberrant, giving rise to a maladaptive fibrotic scar or adipose tissue infiltration, mainly due to dysregulated activity of different muscle interstitial cells. In this context, plasma preparations, including Platelet-Rich Plasma (PRP) and more recently Platelet-Poor Plasma (PPP), have shown advantages and promising therapeutic perspectives. This review focuses on the contribution of these blood-derived products on repair/regeneration of damaged skeletal muscle, paying particular attention to the potential cellular targets and molecular mechanisms through which these products may exert their beneficial effects.
Keywords: Platelet-Poor Plasma (PPP); Platelet-Rich Plasma (PRP); fibrosis; myoblasts; myofibroblasts; myogenesis; regenerative medicine; satellite cells; skeletal muscle regeneration; stem cell niche.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Schaaf G., Sage F., Stok M., Brusse E., Pijnappel W.W.M., Reuser A.J., vd Ploeg A.T. Ex-vivo Expansion of Muscle-Regenerative Cells for the Treatment of Muscle Disorders. J. Stem Cell Res. 2012;S11:003. doi: 10.4172/2157-7633.S11-003. - DOI
-
- Sassoli C., Pini A., Chellini F., Mazzanti B., Nistri S., Nosi D., Saccardi R., Quercioli F., Zecchi-Orlandini S., Formigli L. Bone marrow mesenchymal stromal cells stimulate skeletal myoblast proliferation through the paracrine release of VEGF. PLoS ONE. 2012;7:e37512. doi: 10.1371/journal.pone.0037512. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials