Mobile platform for rapid sub-picogram-per-milliliter, multiplexed, digital droplet detection of proteins
- PMID: 30765530
- PMCID: PMC6410864
- DOI: 10.1073/pnas.1814110116
Mobile platform for rapid sub-picogram-per-milliliter, multiplexed, digital droplet detection of proteins
Abstract
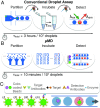
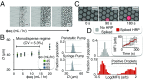
Digital droplet assays-in which biological samples are compartmentalized into millions of femtoliter-volume droplets and interrogated individually-have generated enormous enthusiasm for their ability to detect biomarkers with single-molecule sensitivity. These assays have untapped potential for point-of-care diagnostics but are currently mainly confined to laboratory settings, due to the instrumentation necessary to serially generate, control, and measure tens of millions of droplets/compartments. To address this challenge, we developed an optofluidic platform that miniaturizes digital assays into a mobile format by parallelizing their operation. This technology is based on three key innovations: (i) the integration and parallel operation of a hundred droplet generators onto a single chip that operates >100× faster than a single droplet generator, (ii) the fluorescence detection of droplets at >100× faster than conventional in-flow detection using time domain-encoded mobile phone imaging, and (iii) the integration of on-chip delay lines and sample processing to allow serum-to-answer device operation. To demonstrate the power of this approach, we performed a duplex digital ELISA. We characterized the performance of this assay by first using spiked recombinant proteins in a complex media (FBS) and measured a limit of detection, 0.004 pg/mL (300 aM), a 1,000× improvement over standard ELISA and matching that of the existing laboratory-based gold standard digital ELISA system. We additionally measured endogenous GM-CSF and IL6 in human serum from n = 14 human subjects using our mobile duplex assay, and showed excellent agreement with the gold standard system ([Formula: see text]).
Keywords: ELISA; cell phone; digital; multiplex; portable.
Copyright © 2019 the Author(s). Published by PNAS.
Conflict of interest statement
Conflict of interest statement: D.I. is the founder and D.I. and V.Y. currently hold equity in Chip Diagnostics. J.R.B. is employed by Chip Diagnostics.
Figures




References
-
- Pekin D, et al. Quantitative and sensitive detection of rare mutations using droplet-based microfluidics. Lab A Chip. 2011;11:2156. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

