ERK Inhibitor Enhances Everolimus Efficacy through the Attenuation of dNTP Pools in Renal Cell Carcinoma
- PMID: 30771617
- PMCID: PMC6374702
- DOI: 10.1016/j.omtn.2019.01.001
ERK Inhibitor Enhances Everolimus Efficacy through the Attenuation of dNTP Pools in Renal Cell Carcinoma
Abstract
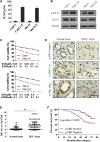
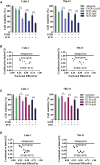
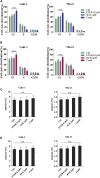
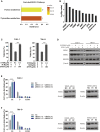
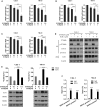
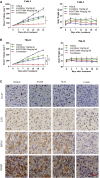
The clinical efficiency of everolimus, an mammalian target of rapamycin (mTOR) inhibitor, is palliative as sequential or second-line therapy for renal cell carcinoma (RCC). However, the limited response of everolimus in RCC remains uncertain. In the present study, everolimus-resistant RCC models were established to understand the mechanisms and to seek combination approaches. Consequently, the activation of ERK was found to contribute toward everolimus-acquired resistance and poor prognosis in patients with RCC. In addition, the efficacy and mechanism of combination treatment underlying RCC using everolimus and ERK inhibitors was investigated. The ERK inhibitor in combination with everolimus synergistically inhibited the proliferation of RCC cells by arresting the cell cycle in the G1 phase. The combination treatment markedly attenuated the deoxyribonucleoside triphosphate (dNTP) pools by downregulating the mRNA expression of RRM1 and RRM2 through E2F1. The overexpression of E2F1 or supplementation of dNTP rescued the anti-proliferation activity of the everolimus-SCH772984 combination. The antitumor efficacy of combination therapy was reiterated in RCC xenograft models. Thus, the current findings provided evidence that the everolimus-ERK inhibitor combination is a preclinical therapeutic strategy for RCC.
Keywords: ERK inhibitor; dNTP; everolimus; renal cell carcinoma; ribonucleotide reductase.
Copyright © 2019 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
References
-
- Fisher R., Gore M., Larkin J. Current and future systemic treatments for renal cell carcinoma. Semin. Cancer Biol. 2013;23:38–45. - PubMed
-
- Motzer R.J., Escudier B., Oudard S., Hutson T.E., Porta C., Bracarda S., Grünwald V., Thompson J.A., Figlin R.A., Hollaender N., RECORD-1 Study Group Phase 3 trial of everolimus for metastatic renal cell carcinoma : final results and analysis of prognostic factors. Cancer. 2010;116:4256–4265. - PubMed
-
- Czarnecka A.M., Kornakiewicz A., Lian F., Szczylik C. Future perspectives for mTOR inhibitors in renal cell cancer treatment. Future Oncol. 2015;11:801–817. - PubMed
-
- Motzer R.J., Hutson T.E., Glen H., Michaelson M.D., Molina A., Eisen T., Jassem J., Zolnierek J., Maroto J.P., Mellado B. Lenvatinib, everolimus, and the combination in patients with metastatic renal cell carcinoma: a randomised, phase 2, open-label, multicentre trial. Lancet Oncol. 2015;16:1473–1482. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

