Transcatheter arterial chemoembolisation followed by three-dimensional conformal radiotherapy versus transcatheter arterial chemoembolisation alone for primary hepatocellular carcinoma in adults
- PMID: 30776082
- PMCID: PMC6378926
- DOI: 10.1002/14651858.CD012244.pub2
Transcatheter arterial chemoembolisation followed by three-dimensional conformal radiotherapy versus transcatheter arterial chemoembolisation alone for primary hepatocellular carcinoma in adults
Abstract
Background: Hepatocellular carcinoma, also called malignant hepatoma, is a primary malignancy of the liver. Despite regular surveillance conducted in high-risk populations, most people with hepatocellular carcinoma are diagnosed at an advanced stage. Consequently, only a minority of people with the disease are suitable for surgical resection when diagnosed.
Objectives: To compare the beneficial and harmful effects of transcatheter arterial chemoembolisation (TACE) followed by three-dimensional conformal radiotherapy (3-DCRT) versus TACE alone in adults with primary hepatocellular carcinoma, considered unsuitable for surgical resection.
Search methods: We searched The Cochrane Hepato-Biliary Group Controlled Trials Register, CENTRAL, MEDLINE, Embase, LILACS, Science Citation Index Expanded, and Conference Proceedings Citation Index - Science up to 31 May 2018. We checked reference lists for all included studies and related reviews for further relevant articles.
Selection criteria: We included all randomised clinical trials comparing TACE followed by 3-DCRT versus TACE alone in people with primary hepatocellular carcinoma.
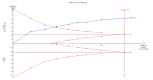
Data collection and analysis: We used standard methodological procedures as suggested by Cochrane. We presented the results of the fixed-effect model in the absence of statistical heterogeneity. Otherwise, we reported the results from the random-effects model meta-analysis. We assessed risk of bias of the included trials using bias risk domains and presented the review results incorporating the methodological quality of the trials using GRADE. Our main conclusions were based on the analysis up to three years' follow-up.
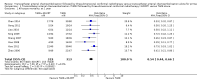
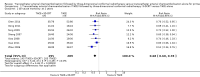
Main results: We identified eight randomised clinical trials (632 participants) that fulfilled our inclusion criteria. All eight trials were at high risk of bias, and we rated the evidence as low to very low certainty. The mean age ranged from 16 years to 78 years. The proportion of men ranged from 60% to 75% and the proportion of people with stage III primary hepatocellular carcinoma ranged from 22% to 85%. The median follow-up duration was 12 months (2 months to 38 months).TACE followed by 3-DCRT compared with TACE alone may have reduced all-cause mortality at three years' follow-up (risk ratio (RR) 0.80, 95% confidence interval (CI) 0.73 to 0.88; 552 participants; 7 trials; low-certainty evidence). TACE followed by 3-DCRT compared with TACE alone may reduce the proportion of participants without tumour response (complete response plus partial response) (RR 0.49, 95% CI 0.39 to 0.61; 632 participants; 8 trials; low-certainty evidence). Data, from one trial on health-related quality of life, favoured the TACE followed by 3-DCRT group, but the provided data were ill-defined (very low-certainty evidence). None of the trials reported serious adverse events. The results on non-serious adverse events were as follows: TACE followed by 3-DCRT compared with TACE alone showed no difference in the results for proportion of participants with leukopenia (RR 1.12, 95% CI 0.92 to 1.34; 438 participants; 5 trials; very low-certainty evidence) and serum transaminases elevation (RR 1.67, 95% CI 0.66 to 4.27; 280 participants; 4 trials; very low-certainty evidence). However, the proportion of participants with total bilirubin elevation was larger in the TACE followed by 3-DCRT group than in the TACE alone group (RR 2.69, 95% CI 1.34 to 5.40; 172 participants; 2 trials; very low-certainty evidence). The rate of participants with serum alpha-fetoprotein (AFP) without decline or normalisation was significantly lower in the TACE followed by 3-DCRT group than in the TACE group, but these data were from one trial only (Chi² = 7.24, P = 0.007; very low-certainty evidence).
Authors' conclusions: TACE followed by 3-DCRT may be associated with lower all-cause mortality and increased tumour response, despite the increased toxicity expressed by a higher rise of total bilirubin. Our review findings should be considered with caution because of the methodological weaknesses in the included trials, resulting in low- to very low-certainty evidence. Data on serious adverse events and health-related quality of life are lacking. We are also very much uncertain in the results of the reported non-serious adverse events. High-quality trials are needed to assess further the role of TACE followed by 3-DCRT for unresectable hepatocellular carcinoma.
Conflict of interest statement
LL: none ZJ: none WZ: none CT: none NX: none
Figures

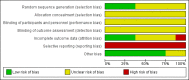
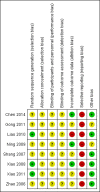
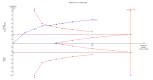

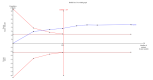









Update of
References
References to studies included in this review
Chen 2014 {published data only}
-
- Chen WJ, Yuan SF, Zhu LJ, Sun XN, Zheng W. Three‐dimensional conformal radiotherapy in combination with transcatheter arterial chemoembolization in the treatment of hepatocellular carcinoma. Journal of B.U.ON. 2014;19(3):692‐7. - PubMed
Gong 2011 {published data only}
-
- Gong QY, Li JG, Kang GL. Effects of transcatheter arterial chemoembolization (TACE) versus TACE combined with three dimensional radiation (3‐DCRT) in the treatment of patients with primary hepatocellular carcinoma. Practical Journal of Cancer 2011;26(6):634‐6.
Liao 2010 {published data only}
-
- Liao XF, He HJ, Zhou ZS, Hu W, Zhu XP. Three dimensional conformal radiotherapy combined with interventional therapy for primary hepatocellular carcinoma. Journal of Practical Oncology 2010;25(6):681‐4.
Ning 2009 {published data only}
-
- Ning SH, Li GF, Huang YS, Li GS. Three‐dimensional conformal radiotherapy combined with transcatheter arterial chemoembolization for hepatocellular carcinoma. Sichuan Medical Journal 2009;30(12):1896‐8.
Shang 2007 {published data only}
-
- Shang Y, You GX, Xu HY, Chen MC. Prospective randomised clinical study of transcatheter arterial chemoembolization, combined with three‐dimensional conformal radiotherapy for primary liver cancer: an analysis of 40 cases. World Chinese Journal of Digestology 2007;15(29):3140‐2.
Xiao 2008 {published data only}
-
- Xiao ZM, Ouyang TC, Yu RZ, Jiang XS, Ren H, Wu ZJ. Transcatheter arterial chemoembolization combined with 3‐dimensional conformal radiotherapy for patients with unresectable primary hepatic carcinoma. Chinese Journal of Clinical Oncology 2008;35(1):18‐21.
Xiao 2011 {published data only}
-
- Xiao P, Dong Y, Sun Y, Liu QF, Cao HM, Xu ZY, et al. Effect comparison of TACE plus 3DCRT and TACE alone in the treatment of primary hepatic cancer. Journal of Basic and Clinical Oncology 2011;24(6):506‐7.
Zhao 2006 {published data only}
-
- Zhao MH, Lang FP, Jiang QA, Ma JJ, Song YX. Three‐dimensional conformal radiotherapy combined with transcatheter arterial chemoembolization for inoperable primary liver cancer. Chinese Journal of Radiation Oncology 2006;15(1):39‐41.
References to studies excluded from this review
Chia‐Hsien 2001 {published data only}
-
- Chia‐Hsien Cheng J, Chuang VP, Cheng SH, Lin YM, Cheng TI, Yang PS, et al. Unresectable hepatocellular carcinoma treated with radiotherapy and/or chemoembolization. International Journal of Cancer 2001;96(4):243‐52. - PubMed
Chung 2006 {published data only}
-
- Chung YL, Jian JJ, Cheng SH, Tsai SY, Chuang VP, Soong T, et al. Sublethal irradiation induces vascular endothelial growth factor and promotes growth of hepatoma cells: implications for radiotherapy of hepatocellular carcinoma. Clinical Cancer Research 2006;12(9):2706‐15. - PubMed
Guo 2005 {published data only}
-
- Guo JX, Sun XN, Huang MX, Chen J, Xu ZY, Ma QB. Evaluation of the efficacy for alternated treatment on primary liver cancer by interventional therapy in combination with fractionated stereotactic conformal radiotherapy. Chinese Journal of Clinical Oncology 2005;32(24):1418‐20.
Koo 2010 {published data only}
-
- Koo JE, Kim JH, Lim YS, Park SJ, Won HJ, Sung KB, et al. Combination of transarterial chemoembolization and three‐dimensional conformal radiotherapy for hepatocellular carcinoma with inferior vena cava tumor thrombus. International Journal of Radiation Oncology, Biology, Physics 2010;78(1):180‐7. - PubMed
Liang 2005 {published data only}
-
- Liang SX, Zhu XD, Lu HJ, Pan CY, Li FX, Huang QF, et al. Hypofractionated three‐dimensional conformal radiation therapy for primary liver carcinoma. Cancer 2005;103(10):2181‐8. - PubMed
Lu 2015 {published data only}
-
- Lu DH, Fei ZL, Zhou JP, Hu ZT, Hao WS. A comparison between three‐dimensional conformal radiotherapy combined with interventional treatment and interventional treatment alone for hepatocellular carcinoma with portal vein tumour thrombosis. Journal of Medical Imaging and Radiation Oncology 2015;59(1):109‐14. - PubMed
Shim 2005 {published data only}
-
- Shim SJ, Seong J, Han KH, Chon CY, Suh CO, Lee JT. Local radiotherapy as a complement to incomplete transcatheter arterial chemoembolization in locally advanced hepatocellular carcinoma. Liver International 2005;25(6):1189‐96. - PubMed
You 2007 {published data only}
-
- You CR, Jang JW, Kang SH, Bae SH, Choi JY, Yoon SK, et al. Efficacy of transarterial chemolipiodolization with or without 3‐dimensional conformal radiotherapy for huge HCC with portal vein tumor thrombosis. Korean Journal of Hepatology 2007;13(3):378‐86. - PubMed
Zeng 2004 {published data only}
-
- Zeng ZC, Tang ZY, Fan J, Zhou J, Qin LX, Ye SL, et al. A comparison of chemoembolization combination with and without radiotherapy for unresectable hepatocellular carcinoma. Cancer Journal (Sudbury, Mass.) 2004;10(5):307‐16. - PubMed
Zhang 2009 {published data only}
-
- Zhang XB, Wang JH, Yan ZP, Qian S, Du SS, Zeng ZC. Hepatocellular carcinoma with main portal vein tumor thrombus: treatment with 3‐dimensional conformal radiotherapy after portal vein stenting and transarterial chemoembolization. Cancer 2009;115(6):1245‐52. - PubMed
Additional references
Bai 2016
Balshem 2011
-
- Balshem H, Helfand M, Schünemann HJ, Oxman AD, Kunz R, Brozek J, et al. GRADE guidelines: 3. Rating the quality of evidence. Journal of Clinical Epidemiology 2011;64(4):401‐6. [PUBMED: 21208779] - PubMed
Brok 2008
-
- Brok J, Thorlund K, Gluud C, Wetterslev J. Trial sequential analysis reveals insufficient information size and potentially false positive results in many meta‐analyses. Journal of Clinical Epidemiology 2008;61(8):763‐9. - PubMed
Brok 2009
-
- Brok J, Thorlund K, Wetterslev J, Gluud C. Apparently conclusive meta‐analyses may be inconclusive – trial sequential analysis adjustment of random error risk due to repetitive testing of accumulating data in apparently conclusive neonatal meta‐analyses. International Journal of Epidemiology 2009;38(1):287‐98. - PubMed
Bronowicki 1994
-
- Bronowicki JP, Vetter D, Dumas F, Boudjema K, Bader R, Weiss AM, et al. Transcatheter oily chemoembolization for hepatocellular carcinoma. A 4‐year study of 127 French patients. Cancer 1994;74(1):16‐24. - PubMed
Cabrera 2010
-
- Cabrera R, Nelson DR. Review article: the management of hepatocellular carcinoma. Alimentary Pharmacology & Therapeutics 2010;31(4):461‐76. - PubMed
Cammà 2002
-
- Cammà C, Schepis F, Orlando A, Albanese M, Shahied L, Trevisani F, et al. Transarterial chemoembolization for unresectable hepatocellular carcinoma: meta‐analysis of randomized controlled trials. Radiology 2002;224(1):47‐54. [MEDLINE: ] - PubMed
Castellini 2018
Cha 2005
-
- Cha C, Dematteo RP. Molecular mechanisms in hepatocellular carcinoma development. Best Practice & Research. Clinical Gastroenterology 2005;19(1):25‐37. - PubMed
Cheng 2000
-
- Cheng JC, Chuang VP, Cheng SH, Huang AT, Lin YM, Cheng TI, et al. Local radiotherapy with or without transcatheter arterial chemoembolization for patients with unresectable hepatocellular carcinoma. International Journal of Radiation Oncology, Biology, Physics 2000;47(2):435‐42. - PubMed
DeMets 1987
-
- DeMets DL. Methods for combining randomized clinical trials: strengths and limitations. Statistics in Medicine 1987;6(3):341‐50. [MEDLINE: ] - PubMed
DerSimonian 1986
-
- DerSimonian R, Laird N. Meta‐analysis in clinical trials. Controlled Clinical Trials 1986;7(3):177‐88. [MEDLINE: ] - PubMed
Deuffic 1998
-
- Deuffic S, Poynard T, Buffat L, Valleron AJ. Trends in primary liver cancer. Lancet 1998;351(9097):214‐5. - PubMed
Egger 1997
El‐Serag 2001
-
- El‐Serag HB. Epidemiology of hepatocellular carcinoma. Clinics in Liver Disease 2001;5(1):87‐107, vi. - PubMed
El‐Serag 2008
-
- El‐Serag HB, Marrero JA, Rudolph L, Reddy KR. Diagnosis and treatment of hepatocellular carcinoma. Gastroenterology 2008;134(6):1752‐63. - PubMed
El‐Serag 2014
Feng 2011
-
- Feng M, Ben‐Josef E. Radiation therapy for hepatocellular carcinoma. Seminars in Radiation Oncology 2011;21(4):271‐7. - PubMed
Ferlay 2015
-
- Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. International Journal of Cancer. Journal International du Cancer 2015;136(5):E359‐86. - PubMed
GRADEpro [Computer program]
-
- Brozek J, Oxman A, Schünemann H. GRADEpro. Version 3.2 for Windows. Grade Working Group 2004‐2007, 2008.
Guyatt 2011a
-
- Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction – GRADE evidence profiles and summary of findings tables. Journal of Clinical Epidemiology 2011;64(4):383‐94. [PUBMED: 21195583] - PubMed
Guyatt 2011b
-
- Guyatt GH, Oxman AD, Kunz R, Atkins D, Brozek J, Vist G, et al. GRADE guidelines: 2. Framing the question and deciding on important outcomes. Journal of Clinical Epidemiology 2011;64(4):395‐400. [PUBMED: 21194891] - PubMed
Guyatt 2011c
-
- Guyatt GH, Oxman AD, Vist G, Kunz R, Brozek J, Alonso‐Coello P, et al. GRADE guidelines: 4. Rating the quality of evidence – study limitations (risk of bias). Journal of Clinical Epidemiology 2011;64(4):407‐15. [PUBMED: 21247734] - PubMed
Guyatt 2011d
-
- Guyatt GH, Oxman AD, Montori V, Vist G, Kunz R, Brozek J, et al. GRADE guidelines: 5. Rating the quality of evidence – publication bias. Journal of Clinical Epidemiology 2011;64(12):1277‐82. [PUBMED: 21802904] - PubMed
Guyatt 2011e
-
- Guyatt GH, Oxman AD, Kunz R, Brozek J, Alonso‐Coello P, Rind D, et al. GRADE guidelines: 6. Rating the quality of evidence – imprecision. Journal of Clinical Epidemiology 2011;64(12):1283‐93. [PUBMED: 21839614] - PubMed
Guyatt 2011f
-
- Guyatt GH, Oxman AD, Kunz R, Woodcock J, Brozek J, Helfand M, et al. GRADE guidelines: 7. Rating the quality of evidence – inconsistency. Journal of Clinical Epidemiology 2011;64(12):1294‐302. [PUBMED: 21803546] - PubMed
Guyatt 2011g
-
- Guyatt GH, Oxman AD, Kunz R, Woodcock J, Brozek J, Helfand M, et al. GRADE guidelines: 8. Rating the quality of evidence – indirectness. Journal of Clinical Epidemiology 2011;64(12):1303‐10. [PUBMED: 21802903] - PubMed
Guyatt 2011h
-
- Guyatt GH, Oxman AD, Sultan S, Glasziou P, Akl EA, Alonso‐Coello P, et al. GRADE guidelines: 9. Rating up the quality of evidence. Journal of Clinical Epidemiology 2011;64(12):1311‐6. [PUBMED: 21802902] - PubMed
Guyatt 2013a
-
- Guyatt G, Oxman AD, Sultan S, Brozek J, Glasziou P, Alonso‐Coello P, et al. GRADE guidelines: 11. Making an overall rating of confidence in effect estimates for a single outcome and for all outcomes. Journal of Clinical Epidemiology 2013;66(2):151‐7. [PUBMED: 22542023] - PubMed
Guyatt 2013b
-
- Guyatt GH, Oxman AD, Santesso N, Helfand M, Vist G, Kunz R, et al. GRADE guidelines: 12. Preparing summary of findings tables – binary outcomes. Journal of Clinical Epidemiology 2013;66(2):158‐72. [PUBMED: 22609141] - PubMed
Guyatt 2013c
-
- Guyatt GH, Thorlund K, Oxman AD, Walter SD, Patrick D, Furukawa TA, et al. GRADE guidelines: 13. Preparing summary of findings tables and evidence profiles – continuous outcomes. Journal of Clinical Epidemiology 2013;66(2):173‐83. [PUBMED: 23116689] - PubMed
Guyatt 2013d
-
- Guyatt G, Andrews J, Oxman AD, Alderson P, Dahm P, Falck‐Ytter Y, et al. GRADE guidelines: 15. Going from evidence to recommendations: the significance and presentation of recommendations. Journal of Clinical Epidemiology 2013;66(7):719‐25. - PubMed
Guyatt 2017
-
- Guyatt GH, Ebrahim S, Alonso‐Coello P, Johnston BC, Mathioudakis AG, Briel M, et al. GRADE guidelines 17: assessing the risk of bias associated with missing participant outcome data in a body of evidence. Journal of Clinical Epidemiology 2017;87:14‐22. [PUBMED: 28529188] - PubMed
Higgins 2002
-
- Higgins JP, Thompson SG. Quantifying heterogeneity in a meta‐analysis. Statistics in Medicine 2002;21(11):1539‐58. [MEDLINE: ] - PubMed
Higgins 2011
-
- Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
ICH‐GCP 1997
-
- International Conference on Harmonisation Expert Working Group. International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use. ICH harmonised tripartite guideline. Guideline for good clinical practice CFR & ICH Guidelines. Vol. 1, Philadelphia (PA): Barnett International/PAREXEL, 1997.
Ikeda 1991
-
- Ikeda K, Kumada H, Saitoh S, Arase Y, Chayama K. Effect of repeated transcatheter arterial embolization on the survival time in patients with hepatocellular carcinoma. An analysis by the Cox proportional hazard model. Cancer 1991;68(10):2150‐4. - PubMed
Jakobsen 2014
Kjaergard 2001
-
- Kjaergard LL, Villumsen J, Gluud C. Reported methodologic quality and discrepancies between large and small randomized trials in meta‐analyses. Annals of Internal Medicine 2001;135(11):982‐9. - PubMed
Kothary 2007
-
- Kothary N, Weintraub JL, Susman J, Rundback JH. Transarterial chemoembolization for primary hepatocellular carcinoma in patients at high risk. Journal of Vascular and Interventional Radiology : JVIR 2007;18(12):1517‐26. [MEDLINE: ] - PubMed
Kumar 2004
-
- Kumar V, Fausto N, Abbas A. Robbins & Cotran Pathologic Basis of Disease. 7th Edition. Philadelphia (PA): Saunders, 2004.
Lan 1983
-
- Lan GK, DeMets GL. Discrete sequential boundaries for clinical trials. Biometrika 1983;70(3):659‐63.
Lawrence 1990
-
- Lawrence TS, Tesser RJ, Haken RK. An application of dose volume histograms to the treatment of intrahepatic malignancies with radiation therapy. International Journal of Radiation Oncology, Biology, Physics 1990;19(4):1041‐7. [MEDLINE: ] - PubMed
Lee 2013
Li 2013
-
- Li SH, Guo ZX, Xiao CZ, Wei W, Shi M, Chen ZY, et al. Risk factors for early and late intrahepatic recurrence in patients with single hepatocellular carcinoma without macrovascular invasion after curative resection. Asian Pacific Journal of Cancer Prevention 2013;14(8):4759‐63. - PubMed
Liang 2015
-
- Liang HY, Guo QY, Sun W, Mao XN, Wen F, Shan M, et al. Sequential use of transhepatic arterial chemoembolization and bipolar radiofrequency ablation in the clinical therapy of hepatocellular carcinoma. Cancer Biotherapy and Radiopharmaceuticals 2015;30(10):427‐32. - PubMed
Llovet 2002
-
- Llovet JM, Real MI, Montaña X, Planas R, Coll S, Aponte J, et al. Arterial embolization or chemoembolization versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet 2002;359(9319):1734‐9. [MEDLINE: ] - PubMed
Llovet 2003
-
- Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: chemoembolization improves survival. Hepatology (Baltimore, Md.) 2003;37(2):429‐42. [MEDLINE: ] - PubMed
Llovet 2004
-
- Llovet JM, Fuster J, Bruix J. The Barcelona approach: diagnosis, staging, and treatment of hepatocellular carcinoma. Liver Transplantation 2004;10:S115‐20. [MEDLINE: ] - PubMed
Lo 2002
-
- Lo CM, Ngan H, Tso WK, Liu CL, Lam CM, Poon RT, et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology (Baltimore, Md.) 2002;35(5):1164‐71. [MEDLINE: ] - PubMed
Lodato 2006
Lundh 2017
Macaskill 2001
-
- Macaskill P, Walter SD, Irwig L. A comparison of methods to detect publication bias in meta‐analysis. Statistics in Medicine 2001;20(4):641‐54. [MEDLINE: ] - PubMed
Matsuura 1998
-
- Matsuura M, Nakajima N, Arai K, Ito K. The usefulness of radiation therapy for hepatocellular carcinoma. Hepato‐Gastroenterology 1998;45(21):791‐6. [MEDLINE: ] - PubMed
McGlynn 2001
-
- McGlynn KA, Tsao L, Hsing AW, Devesa SS, Fraumeni JF Jr. International trends and patterns of primary liver cancer. International Journal of Cancer. Journal International du Cancer 2001;94(2):290‐6. - PubMed
Mittal 2013
Moher 1998
-
- Moher D, Pham B, Jones A, Cook DJ, Jadad AR, Moher M, et al. Does quality of reports of randomised trials affect estimates of intervention efficacy reported in meta‐analyses?. Lancet 1998;352(9128):609‐13. - PubMed
Murata 2014
Mustafa 2013
-
- Mustafa RA, Santesso N, Brozek J, Akl EA, Walter SD, Norman G, et al. The GRADE approach is reproducible in assessing the quality of evidence of quantitative evidence syntheses. Journal of Clinical Epidemiology 2013;66(7):736‐42; quiz 742.e1‐5. [PUBMED: 23623694] - PubMed
Nakamura 1990
-
- Nakamura H, Hashimoto T, Oi H, Sawada S, Furui S, Mizumoto S, et al. Treatment of hepatocellular carcinoma by segmental hepatic artery injection of adriamycin‐in‐oil emulsion with overflow to segmental portal veins. Acta Radiologica 1990;31(4):347‐9. - PubMed
Oliveri 2011
Park 2014
-
- Park ES, Kwon do H, Park JB, Lee do H, Cho YH, Kim JH, et al. Gamma Knife surgery for treating brain metastases arising from hepatocellular carcinomas. Journal of Neurosurgery 2014;121 Suppl:102‐9. - PubMed
Review Manager 2014 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Robertson 1993
-
- Robertson JM, Lawrence TS, Dworzanin LM, Andrews JC, Walker S, Kessler ML, et al. Treatment of primary hepatobiliary cancers with conformal radiation therapy and regional chemotherapy. Journal of Clinical Oncology 1993;11(7):1286‐93. [MEDLINE: ] - PubMed
Royle 2003
-
- Royle P, Milne R. Literature searching for randomized controlled trials used in Cochrane reviews: rapid versus exhaustive searches. International Journal of Technology Assessment in Health Care 2003;19(4):591‐603. [MEDLINE: ] - PubMed
Sasaki 1987
-
- Sasaki Y, Imaoka S, Kasugai H, Fujita M, Kawamoto S, Ishiguro S, et al. A new approach to chemoembolization therapy for hepatoma using ethiodized oil, cisplatin, and gelatin sponge. Cancer 1987;60(6):1194‐203. [MEDLINE: ] - PubMed
Savović 2012a
-
- Savović J, Jones HE, Altman DG, Harris RJ, Jüni P, Pildal J, et al. Influence of reported study design characteristics on intervention effect estimates from randomized, controlled trials. Health Technology Assessment 2012;16(35):1‐82. - PubMed
Savović 2012b
-
- Savović J, Jones HE, Altman DG, Harris RJ, Jüni P, Pildal J, et al. Influence of reported study design characteristics on intervention effect estimates from randomized, controlled trials. Annals of Internal Medicine 2012;157(6):429‐38. - PubMed
Savović 2018
Schulz 1995
-
- Schulz KF, Chalmers I, Hayes RJ, Altman DG. Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials. JAMA 1995;273(5):408‐12. - PubMed
Seong 2000
-
- Seong J, Park HC, Han KH, Lee DY, Lee JT, Chon CY, et al. Local radiotherapy for unresectable hepatocellular carcinoma patients who failed with transcatheter arterial chemoembolization. International Journal of Radiation Oncology, Biology, Physics 2000;47(5):1331‐5. - PubMed
Spieth 2003
-
- Spieth K, Kaufmann R, Gille J. Metronomic oral low‐dose treosulfan chemotherapy combined with cyclooxygenase‐2 inhibitor in pretreated advanced melanoma: a pilot study. Cancer Chemotherapy and Pharmacology 2003;52(5):377‐82. - PubMed
Storebø 2018
-
- Storebø OJ, Pedersen N, Ramstad E, Kielsholm ML, Nielsen SS, Krogh HB, et al. Methylphenidate for attention deficit hyperactivity disorder (ADHD) in children and adolescents – assessment of adverse events in non‐randomised studies. Cochrane Database of Systematic Reviews 2018, Issue 5. [DOI: 10.1002/14651858.CD012069.pub2] - DOI - PMC - PubMed
Takayasu 1987
-
- Takayasu K, Shima Y, Muramatsu Y, Moriyama N, Yamada T, Makuuchi M, et al. Hepatocellular carcinoma: treatment with intraarterial iodized oil with and without chemotherapeutic agents. Radiology 1987;163(2):345‐51. - PubMed
Takayasu 2006
-
- Takayasu K, Arii S, Ikai I, Omata M, Okita K, Ichida T, et al. Prospective cohort study of transarterial chemoembolization for unresectable hepatocellular carcinoma in 8510 patients. Gastroenterology 2006;131(2):461‐9. [MEDLINE: ] - PubMed
Thorlund 2009
-
- Thorlund K, Devereaux PJ, Wetterslev J, Guyatt G, Ioannidis JP, Thabane L, et al. Can trial sequential monitoring boundaries reduce spurious inferences from meta‐analyses?. International Journal of Epidemiology 2009;38(1):276‐86. - PubMed
Thorlund 2010
Thorlund 2011
-
- Thorlund K, Engstrøm J, Wetterslev J, Brok J, Imberger G, Gluud C. User manual for Trial Sequential Analysis (TSA), 2011. ctu.dk/tsa/files/tsa_manual.pdf (accessed 27 May 2017).
TSA 2011 [Computer program]
-
- Copenhagen Trial Unit. TSA – Trial Sequential Analysis. Version 0.9.5.10 Beta. Copenhagen: Copenhagen Trial Unit, 2011.
Venook 2010
-
- Venook AP, Papandreou C, Furuse J, Guevara LL. The incidence and epidemiology of hepatocellular carcinoma: a global and regional perspective. Oncologist 2010;15 Suppl 4:5‐13. - PubMed
Wetterslev 2008
-
- Wetterslev J, Thorlund K, Brok J, Gluud C. Trial sequential analysis may establish when firm evidence is reached in cumulative meta‐analysis. Journal of Clinical Epidemiology 2008;61(1):64‐75. - PubMed
Wetterslev 2009
Wetterslev 2017
WHO 2009
-
- World Health Organization. Cancer. Fact sheet N°297, 2009. www.who.int/mediacentre/factsheets/fs297/en/ (accessed 30 June 2015).
Wood 2008
Yamagami 2014
-
- Yamagami T, Yoshimatsu R, Ishikawa M, Kajiwara K, Aikata H, Tashiro H, et al. Transcatheter arterial chemoembolization with an interventional‐CT system for recurrent hepatocellular carcinoma after living donor liver transplantation. Hepato‐gastroenterology 2014;61(133):1387‐92. - PubMed
Yoshikawa 1994
-
- Yoshikawa M, Saisho H, Ebara M, Iijima T, Iwama S, Endo F, et al. A randomized trial of intrahepatic arterial infusion of 4'‐epidoxorubicin with Lipiodol versus 4'‐epidoxorubicin alone in the treatment of hepatocellular carcinoma. Cancer Chemotherapy and Pharmacology 1994;33 Suppl:S149‐52. - PubMed
Yu 2014
-
- Yu JI, Park HC. Considerations for radiation therapy in hepatocellular carcinoma: the radiation oncologists' perspective. Digestive Diseases (Basel, Switzerland) 2014;32(6):755‐63. - PubMed
Zhou 2007
-
- Zhou ZH, Liu LM, Chen WW, Men ZQ, Lin JH, Chen Z, et al. Combined therapy of transcatheter arterial chemoembolisation and three‐dimensional conformal radiotherapy for hepatocellular carcinoma. British Journal of Radiology 2007;80(951):194‐201. - PubMed
References to other published versions of this review
Lu 2016
-
- Lu L, Zeng J, Wen Z. Combination of three‐dimensional conformal radiotherapy and transcatheter arterial chemoembolisation versus transcatheter arterial chemoembolisation for primary hepatocellular carcinoma. Cochrane Database of Systematic Reviews 2016, Issue 6. [DOI: 10.1002/14651858.CD012244] - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

