Quantitative and Digital Droplet-Based AAV Genome Titration
- PMID: 30783968
- PMCID: PMC7377043
- DOI: 10.1007/978-1-4939-9139-6_4
Quantitative and Digital Droplet-Based AAV Genome Titration
Abstract
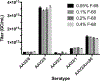
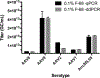
The adeno-associated viral vector (AAV) platform has developed into a primary modality for efficient in vivo, and in more limited settings, in vitro or ex vivo gene transfer. Its applications range from a tool for experimental purposes to preclinical and clinical gene therapy. The ability to accurately and reproducibly quantify vector concentration is critical for any of these applications. While several quantification assays are available, here we outline a detailed protocol for the quantification of DNase-I protected vector genomes reliant on the polymerase chain reaction (PCR) as a measure of the active component of the vector, namely its transgene cargo. With the emergence of droplet digital PCR (ddPCR), we provide side-by-side protocols for traditional TaqMan™ real-time, quantitative PCR (qPCR) and ddPCR, as well as comparative data generated with both methods. Lastly, we discuss the importance of the use of surfactant (here, Pluronic® F-68) in the execution of the assay to limit DNA and AAV adherence to various carriers during the titration, particularly at low concentrations. We believe these protocols can lead to reduced variability and increased comparability between AAV studies.
Keywords: AAV; Adeno-associated virus; Droplet digital PCR; Genome; Quantitative PCR; Real-time PCR; Titration; Vector; ddPCR; qPCR.
Conflict of interest statement
Figures









Similar articles
-
Absolute determination of single-stranded and self-complementary adeno-associated viral vector genome titers by droplet digital PCR.Hum Gene Ther Methods. 2014 Apr;25(2):115-25. doi: 10.1089/hgtb.2013.131. Epub 2014 Feb 14. Hum Gene Ther Methods. 2014. PMID: 24328707 Free PMC article.
-
Two-Dimensional Droplet Digital PCR as a Tool for Titration and Integrity Evaluation of Recombinant Adeno-Associated Viral Vectors.Hum Gene Ther Methods. 2019 Aug;30(4):127-136. doi: 10.1089/hgtb.2019.031. Epub 2019 Jun 21. Hum Gene Ther Methods. 2019. PMID: 31140327 Free PMC article.
-
Accurate Quantification of AAV Vector Genomes by Quantitative PCR.Genes (Basel). 2021 Apr 19;12(4):601. doi: 10.3390/genes12040601. Genes (Basel). 2021. PMID: 33921790 Free PMC article.
-
Expressing Transgenes That Exceed the Packaging Capacity of Adeno-Associated Virus Capsids.Hum Gene Ther Methods. 2016 Feb;27(1):1-12. doi: 10.1089/hgtb.2015.140. Hum Gene Ther Methods. 2016. PMID: 26757051 Free PMC article. Review.
-
From virus evolution to vector revolution: use of naturally occurring serotypes of adeno-associated virus (AAV) as novel vectors for human gene therapy.Curr Gene Ther. 2003 Aug;3(4):281-304. doi: 10.2174/1566523034578285. Curr Gene Ther. 2003. PMID: 12871018 Review.
Cited by
-
An amplification-free CRISPR-Cas12a assay for titer determination and composition analysis of the rAAV genome.Mol Ther Methods Clin Dev. 2024 Jul 22;32(3):101304. doi: 10.1016/j.omtm.2024.101304. eCollection 2024 Sep 12. Mol Ther Methods Clin Dev. 2024. PMID: 39193315 Free PMC article.
-
AAV6 Is Superior to Clade F AAVs in Stimulating Homologous Recombination-Based Genome Editing in Human HSPCs.Mol Ther. 2019 Oct 2;27(10):1701-1705. doi: 10.1016/j.ymthe.2019.09.005. Epub 2019 Sep 12. Mol Ther. 2019. PMID: 31537456 Free PMC article. No abstract available.
-
High-efficiency base editing in the retina in primates and human tissues.Nat Med. 2025 Feb;31(2):490-501. doi: 10.1038/s41591-024-03422-8. Epub 2025 Jan 8. Nat Med. 2025. PMID: 39779923 Free PMC article.
-
Analytical methods to characterize recombinant adeno-associated virus vectors and the benefit of standardization and reference materials.Curr Opin Biotechnol. 2021 Oct;71:65-76. doi: 10.1016/j.copbio.2021.06.025. Epub 2021 Jul 15. Curr Opin Biotechnol. 2021. PMID: 34273809 Free PMC article. Review.
-
Development of a one-step RT-ddPCR method to determine the expression and potency of AAV vectors.Mol Ther Methods Clin Dev. 2021 May 14;23:68-77. doi: 10.1016/j.omtm.2021.05.003. eCollection 2021 Dec 10. Mol Ther Methods Clin Dev. 2021. PMID: 34589555 Free PMC article.
References
-
- Gao G, Vandenberghe LH, Wilson JM (2005) New recombinant serotypes of AAV vectors. Curr Gene Ther 5(3):285–297 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

