Memantine protects thalamocortical hyper-glutamatergic transmission induced by NMDA receptor antagonism via activation of system xc<sup/>
- PMID: 30784207
- PMCID: PMC6323135
- DOI: 10.1002/prp2.457
Memantine protects thalamocortical hyper-glutamatergic transmission induced by NMDA receptor antagonism via activation of system xc<sup/>
Abstract
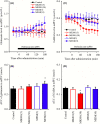
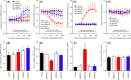
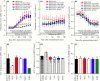
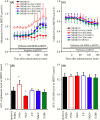
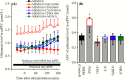
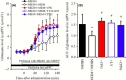
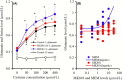
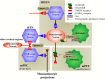
Deficiencies in N-methyl-d-aspartate (NMDA)/glutamate receptor (NMDAR) signaling have been considered central to the cognitive impairments of schizophrenia; however, an NMDAR antagonist memantine (MEM) improves cognitive impairments of Alzheimer's disease and schizophrenia. These mechanisms of paradoxical clinical effects of NMDAR antagonists remain unclear. To explore the mechanisms by which MK801 and MEM affect thalamocortical transmission, we determined interactions between local administrations of MK801, MEM, system xc- (Sxc), and metabotropic glutamate receptors (mGluRs) on extracellular glutamate and GABA levels in the mediodorsal thalamic nucleus (MDTN) and medial prefrontal cortex (mPFC) using dual-probe microdialysis with ultra-high-pressure liquid chromatography. Effects of MK801 and MEM on Sxc activity were also determined using primary cultured astrocytes. Sxc activity was enhanced by MEM, but was unaffected by MK801. MK801 enhanced thalamocortical glutamatergic transmission by GABAergic disinhibition in the MDTN. In the MDTN and the mPFC, MEM weakly increased glutamate release by activating Sxc, whereas MEM inhibited thalamocortical glutamatergic transmission. Paradoxical effects of MEM were induced following secondary activation of inhibitory II-mGluR and III-mGluR by exporting glutamate from astroglial Sxc. The present results suggest that the effects of therapeutically relevant concentrations of MEM on thalamocortical glutamatergic transmission are predominantly caused by activation of Sxc rather than inhibition of NMDAR. These demonstrations suggest that the combination between reduced NMDAR and activated Sxc contribute to the neuroprotective effects of MEM. Furthermore, activation of Sxc may compensate for the cognitive impairments that are induced by hyperactivation of thalamocortical glutamatergic transmission following activation of Sxc/II-mGluR in the MDTN and Sxc/II-mGluR/III-mGluR in the mPFC.
Keywords: cystine/glutamate antiporter; mediodorsal thalamic nucleus; memantine; schizophrenia.
© 2019 The Authors. Pharmacology Research & Perspectives published by John Wiley & Sons Ltd, British Pharmacological Society and American Society for Pharmacology and Experimental Therapeutics.
Conflict of interest statement
The authors state no conflict of interest.
Figures


Similar articles
-
Cystine/Glutamate Antiporter and Aripiprazole Compensate NMDA Antagonist-Induced Dysfunction of Thalamocortical L-Glutamatergic Transmission.Int J Mol Sci. 2018 Nov 19;19(11):3645. doi: 10.3390/ijms19113645. Int J Mol Sci. 2018. PMID: 30463253 Free PMC article.
-
Clozapine Normalizes a Glutamatergic Transmission Abnormality Induced by an Impaired NMDA Receptor in the Thalamocortical Pathway via the Activation of a Group III Metabotropic Glutamate Receptor.Biomolecules. 2019 Jun 17;9(6):234. doi: 10.3390/biom9060234. Biomolecules. 2019. PMID: 31213006 Free PMC article.
-
Pharmacological Discrimination of Effects of MK801 on Thalamocortical, Mesothalamic, and Mesocortical Transmissions.Biomolecules. 2019 Nov 18;9(11):746. doi: 10.3390/biom9110746. Biomolecules. 2019. PMID: 31752219 Free PMC article.
-
Unraveling monoamine receptors involved in the action of typical and atypical antipsychotics on glutamatergic and serotonergic transmission in prefrontal cortex.Curr Pharm Des. 2010;16(5):502-15. doi: 10.2174/138161210790361416. Curr Pharm Des. 2010. PMID: 19909228 Review.
-
Memantine: targeting glutamate excitotoxicity in Alzheimer's disease and other dementias.Am J Alzheimers Dis Other Demen. 2005 Mar-Apr;20(2):77-85. doi: 10.1177/153331750502000206. Am J Alzheimers Dis Other Demen. 2005. PMID: 15844753 Free PMC article. Review.
Cited by
-
Amantadine Improves Delayed Neuropsychiatric Sequelae of Carbon Monoxide Poisoning: A Case Report.Brain Sci. 2019 Oct 25;9(11):292. doi: 10.3390/brainsci9110292. Brain Sci. 2019. PMID: 31731567 Free PMC article.
-
Pathogenesis and pathophysiology of autosomal dominant sleep-related hypermotor epilepsy with S284L-mutant α4 subunit of nicotinic ACh receptor.Br J Pharmacol. 2020 May;177(9):2143-2162. doi: 10.1111/bph.14974. Epub 2020 Feb 15. Br J Pharmacol. 2020. PMID: 31901135 Free PMC article.
-
Vortioxetine Subchronically Activates Serotonergic Transmission via Desensitization of Serotonin 5-HT1A Receptor with 5-HT3 Receptor Inhibition in Rats.Int J Mol Sci. 2019 Dec 10;20(24):6235. doi: 10.3390/ijms20246235. Int J Mol Sci. 2019. PMID: 31835640 Free PMC article.
-
Relationships between Expenditure of Regional Governments and Suicide Mortalities Caused by Six Major Motives in Japan.Int J Environ Res Public Health. 2021 Dec 22;19(1):84. doi: 10.3390/ijerph19010084. Int J Environ Res Public Health. 2021. PMID: 35010343 Free PMC article.
-
Therapeutic Potential and Limitation of Serotonin Type 7 Receptor Modulation.Int J Mol Sci. 2023 Jan 20;24(3):2070. doi: 10.3390/ijms24032070. Int J Mol Sci. 2023. PMID: 36768393 Free PMC article. Review.
References
-
- Malhotra AK, Pinals DA, Adler CM, et al. Ketamine‐induced exacerbation of psychotic symptoms and cognitive impairment in neuroleptic‐free schizophrenics. Neuropsychopharmacology. 1997;17:141‐150. - PubMed
-
- Krystal JH, D'Souza DC, Mathalon D, Perry E, Belger A, Hoffman R. NMDA receptor antagonist effects, cortical glutamatergic function, and schizophrenia: toward a paradigm shift in medication development. Psychopharmacology. 2003;169:215‐233. - PubMed
-
- Singh SP, Singh V. Meta‐analysis of the efficacy of adjunctive NMDA receptor modulators in chronic schizophrenia. CNS Drugs. 2011;25:859‐885. - PubMed
-
- Kishi T, Iwata N. NMDA receptor antagonists interventions in schizophrenia: meta‐analysis of randomized, placebo‐controlled trials. J Psychiatr Res. 2013;47:1143‐1149. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

