Evaluation of hemostasis in patients with end-stage renal disease
- PMID: 30785941
- PMCID: PMC6382154
- DOI: 10.1371/journal.pone.0212237
Evaluation of hemostasis in patients with end-stage renal disease
Abstract
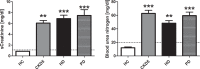
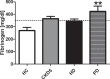
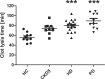
An increased bleeding risk is reported for patients with end-stage renal disease. This study aims to analyze, whether bleeding risk can be assessed by global tests of hemostasis. Standard laboratory tests and an extended evaluation of hemostasis by rotational thromboelastometry, platelet function analyzer (PFA) and multiple electrode aggregometry as well as thrombin generation assays and measurement of fibrinolytic potential were performed in 20 patients on hemodialysis, 10 patients on peritoneal dialysis, 10 patients with chronic kidney disease stage G5 (CKD5) and in 10 healthy controls (HC). Hemoglobin was significantly lower in patients with end-stage renal disease versus HC (each p<0.01). Patients on peritoneal dialysis showed increased fibrinogen levels compared to HC (p<0.01), which were also reflected by FIBTEM results (each p<0.05). 41% of hemodialysis patients and 44% of CKD5 patients presented with prolonged PFA-ADP-test (p<0.05), while no patient on peritoneal dialysis and no HC offered this modification. Thrombin generating potential was significantly lower in patients on hemodialysis, while clot lysis time revealed a hypofibrinolytic state in patients on hemo- and peritoneal dialysis compared to HC (p<0.001). In conclusion, patients with end-stage renal disease have complex hemostatic changes with both hyper- and hypocoagulable features, which are dependent on use and type of dialysis. Hypercoagulable features include elevated fibrinogen levels and a hypofibrinolytic state, whereas hypocoagulable features include decreased thrombin generating capacity and platelet dysfunction. Our results may contribute to a more rational approach to hemostatic management in these patients.
Conflict of interest statement
The authors of this manuscript have read the journal´s policy and have the following competing interests: AG reports personal fees from Alexion and Ablynx/Sanofi. AK reports grants and personal fees from Alexion. OW reports grants and personal fees from Alexion and Pfizer. HR, TL and TB declare no conflicts of interest. FHS reports personal fees from CSL Behring, TEM International and Biotest. This does not alter the authors' adherence to PLOS ONE policies on sharing data and materials.
Figures





References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

