The sustained release of basic fibroblast growth factor accelerates angiogenesis and the engraftment of the inactivated dermis by high hydrostatic pressure
- PMID: 30789932
- PMCID: PMC6383993
- DOI: 10.1371/journal.pone.0208658
The sustained release of basic fibroblast growth factor accelerates angiogenesis and the engraftment of the inactivated dermis by high hydrostatic pressure
Abstract
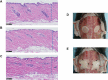
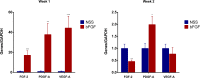
We developed a novel skin regeneration therapy combining nevus tissue inactivated by high hydrostatic pressure (HHP) in the reconstruction of the dermis with a cultured epidermal autograft (CEA). The issue with this treatment is the unstable survival of CEA on the inactivated dermis. In this study, we applied collagen/gelatin sponge (CGS), which can sustain the release of basic fibroblast growth factor (bFGF), to the inactivated skin in order to accelerate angiogenesis. Murine skin grafts from C57BL6J/Jcl mice (8 mm in diameter) were prepared, inactivated by HHP and cryopreserved. One month later, the grafts were transplanted subcutaneously onto the back of other mice and covered by CGS impregnated with saline or bFGF. Grafts were harvested after one, two and eight weeks, at which point the engraftment was evaluated through the histology and angiogenesis-related gene expressions were determined by real-time polymerase chain reaction. Histological sections showed that the dermal cellular density and newly formed capillaries in the bFGF group were significantly higher than in the control group. The relative expression of FGF-2, PDGF-A and VEGF-A genes in the bFGF group was significantly higher than in the control group at Week 1. This study suggested that the angiogenesis into grafts was accelerated, which might improve the engraftment of inactivated dermis in combination with the sustained release of bFGF by CGSs.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
-
- Gospodarowicz D. Localisation of a fibroblast growth factor and its effect alone and with hydrocortisone on 3T3 cell growth. Nature. 1974;249(453):123–7. Epub 1974/05/10. . - PubMed
-
- Hayek A, Culler FL, Beattie GM, Lopez AD, Cuevas P, Baird A. An in vivo model for study of the angiogenic effects of basic fibroblast growth factor. Biochem Biophys Res Commun. 1987;147(2):876–80. Epub 1987/09/15. . - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

