Identification of Broad-Spectrum Antiviral Compounds by Targeting Viral Entry
- PMID: 30791609
- PMCID: PMC6410080
- DOI: 10.3390/v11020176
Identification of Broad-Spectrum Antiviral Compounds by Targeting Viral Entry
Abstract
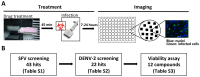
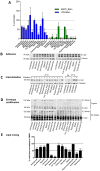
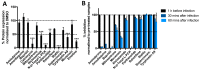
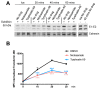
Viruses are a major threat to human health and economic well-being. In recent years Ebola, Zika, influenza, and chikungunya virus epidemics have raised awareness that infections can spread rapidly before vaccines or specific antagonists can be made available. Broad-spectrum antivirals are drugs with the potential to inhibit infection by viruses from different groups or families, which may be deployed during outbreaks when specific diagnostics, vaccines or directly acting antivirals are not available. While pathogen-directed approaches are generally effective against a few closely related viruses, targeting cellular pathways used by multiple viral agents can have broad-spectrum efficacy. Virus entry, particularly clathrin-mediated endocytosis, constitutes an attractive target as it is used by many viruses. Using a phenotypic screening strategy where the inhibitory activity of small molecules was sequentially tested against different viruses, we identified 12 compounds with broad-spectrum activity, and found a subset blocking viral internalisation and/or fusion. Importantly, we show that compounds identified with this approach can reduce viral replication in a mouse model of Zika infection. This work provides proof of concept that it is possible to identify broad-spectrum inhibitors by iterative phenotypic screenings, and that inhibition of host-pathways critical for viral life cycles can be an effective antiviral strategy.
Keywords: Semliki Forest virus; Zika; alphaviruses; broad-spectrum antivirals; dengue; endocytosis; flaviviruses; host-targeted antivirals; phenotypic screening; virus entry.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

