Trends in Human Papillomavirus Vaccine Types 16 and 18 in Cervical Precancers, 2008-2014
- PMID: 30792242
- PMCID: PMC6526945
- DOI: 10.1158/1055-9965.EPI-18-0885
Trends in Human Papillomavirus Vaccine Types 16 and 18 in Cervical Precancers, 2008-2014
Abstract
Background: The impact of human papillomavirus (HPV) vaccination has been observed in the United States through declining cervical precancer incidence in young women. To further evaluate vaccine impact, we described trends in HPV vaccine types 16/18 in cervical precancers, 2008-2014.
Methods: We analyzed data from a 5-site, population-based surveillance system. Archived specimens from women age 18-39 years diagnosed with cervical intraepithelial neoplasia grades 2-3 or adenocarcinoma in situ (CIN2+) were tested for 37 HPV types. We described the proportion and estimated number of cases of CIN2+ by HPV-type groups over time. Trends in HPV16/18-positive CIN2+ were examined, overall and by vaccination status, age, histologic grade, and race/ethnicity, using Cochrane-Armitage tests.
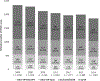
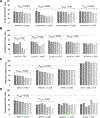
Results: In 10,206 cases, the proportion and estimated number of cases of HPV16/18-positive CIN2+ declined from 52.7% (1,235 cases) in 2008 to 44.1% (819 cases) in 2014 (P < 0.001). Declining trends in the proportion of HPV16/18-positive CIN2+ were observed among vaccinated (55.2%-33.3%, P < 0.001) and unvaccinated (51.0%-47.3%, P = 0.03) women; ages 18-20 (48.7%-18.8%, P = 0.02), 21-24 (53.8%-44.0%, P < 0.001), 25-29 (56.9%-42.4%, P < 0.001), and 30-34 (49.8%-45.8%, P = 0.04) years; CIN2 (40.8%-29.9%, P < 0.001) and CIN2/3 (61.8%-46.2%, P < 0.001); non-Hispanic white (59.5%-47.9%, P < 0.001) and non-Hispanic black (40.7%-26.5%, P < 0.001).
Conclusions: From 2008-2014, the proportion of HPV16/18-positive CIN2+ declined, with the greatest declines in vaccinated women; declines in unvaccinated women suggest herd protection.
Impact: The declining proportion of HPV16/18-positive CIN2+ provides additional evidence of vaccine impact in the United States.
©2019 American Association for Cancer Research.
Conflict of interest statement
Disclosure of Potential Conflicts of Interest
L.M. Niccolai is a consultant/advisory board member for Merck. No potential conflicts of interest were disclosed by the other authors.
Figures
Comment in
-
HPV infections most often linked to cervical cancer have declined.Cancer. 2019 Jul 15;125(14):2336-2337. doi: 10.1002/cncr.32349. Cancer. 2019. PMID: 31260094 No abstract available.
References
-
- Markowitz LE, Dunne EF, Saraiya M, Chesson HW, Curtis CR, Gee J, et al. Human papillomavirus vaccination: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2014;63:1–30. - PubMed

