Retrograde transport of Akt by a neuronal Rab5-APPL1 endosome
- PMID: 30792402
- PMCID: PMC6385319
- DOI: 10.1038/s41598-019-38637-0
Retrograde transport of Akt by a neuronal Rab5-APPL1 endosome
Abstract
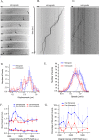
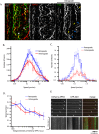
Long-distance axonal trafficking plays a critical role in neuronal function and transport defects have been linked to neurodegenerative disorders. Various lines of evidence suggest that the small GTPase Rab5 plays a role in neuronal signaling via early endosomal transport. Here, we characterized the motility of Rab5 endosomes in primary cultures of mouse hippocampal pyramidal cells by live-cell imaging and showed that they exhibit bi-directional long-range motility in axons, with a strong bias toward retrograde transport. Characterization of key Rab5 effectors revealed that endogenous Rabankyrin-5, Rabenosyn-5 and APPL1 are all present in axons. Further analysis of APPL1-positive endosomes showed that, similar to Rab5-endosomes, they display more frequent long-range retrograde than anterograde movement, with the endosomal levels of APPL1 correlated with faster retrograde movement. Interestingly, APPL1-endosomes transport the neurotrophin receptor TrkB and mediate retrograde axonal transport of the kinase Akt1. FRET analysis revealed that APPL1 and Akt1 interact in an endocytosis-dependent manner. We conclude that Rab5-APPL1 endosomes exhibit the hallmarks of axonal signaling endosomes to transport Akt1 in hippocampal pyramidal cells.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
[Effectors of GTPase Rab5 in endocytosis and signal transduction].Postepy Biochem. 2009;55(2):171-80. Postepy Biochem. 2009. PMID: 19824473 Review. Polish.
-
Evidence that the rab5 effector APPL1 mediates APP-βCTF-induced dysfunction of endosomes in Down syndrome and Alzheimer's disease.Mol Psychiatry. 2016 May;21(5):707-16. doi: 10.1038/mp.2015.97. Epub 2015 Jul 21. Mol Psychiatry. 2016. PMID: 26194181 Free PMC article.
-
Increased Neuronal Expression of the Early Endosomal Adaptor APPL1 Replicates Alzheimer's Disease-Related Endosomal and Synaptic Dysfunction with Cholinergic Neurodegeneration.J Neurosci. 2025 Jul 16;45(29):e2331242025. doi: 10.1523/JNEUROSCI.2331-24.2025. J Neurosci. 2025. PMID: 40514243 Free PMC article.
-
c-Jun N-terminal kinase (JNK)-dependent internalization and Rab5-dependent endocytic sorting mediate long-distance retrograde neuronal death induced by axonal BDNF-p75 signaling.Sci Rep. 2019 Apr 15;9(1):6070. doi: 10.1038/s41598-019-42420-6. Sci Rep. 2019. PMID: 30988348 Free PMC article.
-
APPL1 is a multifunctional endosomal signaling adaptor protein.Biochem Soc Trans. 2017 Jun 15;45(3):771-779. doi: 10.1042/BST20160191. Biochem Soc Trans. 2017. PMID: 28620038 Free PMC article. Review.
Cited by
-
PTPN23-dependent activation of PI3KC2α is a therapeutic vulnerability of BRAF-mutant cancers.J Exp Med. 2025 Mar 3;222(3):e20241147. doi: 10.1084/jem.20241147. Epub 2025 Jan 22. J Exp Med. 2025. PMID: 39841180
-
Axonal transport of Hrs is activity dependent and facilitates synaptic vesicle protein degradation.Life Sci Alliance. 2022 May 30;5(10):e202000745. doi: 10.26508/lsa.202000745. Print 2022 Oct. Life Sci Alliance. 2022. PMID: 35636965 Free PMC article.
-
Direct interaction between Rab5a and Rab4a enhanced epidermal growth factor-stimulated proliferation of gastric cancer cells.World J Gastrointest Oncol. 2021 Oct 15;13(10):1492-1505. doi: 10.4251/wjgo.v13.i10.1492. World J Gastrointest Oncol. 2021. PMID: 34721780 Free PMC article.
-
A germline mutation in Rab43 gene identified from a cancer family predisposes to a hereditary liver-colon cancer syndrome.BMC Cancer. 2019 Jun 21;19(1):613. doi: 10.1186/s12885-019-5845-4. BMC Cancer. 2019. PMID: 31226964 Free PMC article.
-
BDNF/TrkB signaling endosomes in axons coordinate CREB/mTOR activation and protein synthesis in the cell body to induce dendritic growth in cortical neurons.Elife. 2023 Feb 24;12:e77455. doi: 10.7554/eLife.77455. Elife. 2023. PMID: 36826992 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

