Evaluation of accordance of antibiotics package size with recommended treatment duration of guidelines for sore throat and urinary tract infections
- PMID: 30792852
- PMCID: PMC6371554
- DOI: 10.1186/s13756-019-0495-5
Evaluation of accordance of antibiotics package size with recommended treatment duration of guidelines for sore throat and urinary tract infections
Abstract
Background: The aim of this study was to investigate whether marketed antibiotics package sizes are in accordance with treatment durations recommended in guidelines for prescribing antibiotics in sore throat and urinary tract infections.
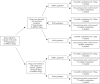
Methods: National drug database was searched with limitation to Antibacterials for systemic use. Formulations which did not have pre-specified dosage unit by the manufacturer were excluded (e.g. powders for oral solutions). The final list contained 94 drugs with 23 different active substances. This list was then cross-referenced with selected antimicrobial prescribing guidelines provided by Intersectoral Society for Antibiotic Resistance Control (ISKRA), National Institute for Health and Care Excellence (NICE) and The Infectious Diseases Society of America (IDSA).
Results: Seven packages matched ISKRA guidelines on sore throat while 16 were mismatched. Considering drug packages under reimbursement, 3 matched ISKRA guidelines and 8 were mismatched. Only 3 packages matched IDSA guidelines for comparable indications, and 18 were mismatched. When considering NICE guidelines there were 5 mismatched and only one package that was in accordance with the guidelines. ISKRA guidelines for urinary tract infections matched 23 packages and mismatched 58 packages. IDSA guidelines for urinary tract infections matched one package and were mismatched in 15 cases.
Conclusions: One of the causes of leftover antibiotics is poor accordance of antibiotic package size with treatment recommendation duration. This should be identified as a potential target for reduction of excess antibiotics in the community. Measures that promote patient adherence to therapy and patient education should be considered essential to manage proper handling of leftover antibiotics.
Keywords: Antibiotic resistance; Prescribing guideline; Sore throat; Urinary tract infections.
Conflict of interest statement
Not applicable.Not applicable.The authors declare that they have no competing interests.Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

References
-
- WHO member states adopt global action plan on antimicrobial resistance. Euro surveill. 2015;20. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

