Interleukin 2 is an Upstream Regulator of CD4+ T Cells From Visceral Leishmaniasis Patients With Therapeutic Potential
- PMID: 30796820
- PMCID: PMC6775044
- DOI: 10.1093/infdis/jiz074
Interleukin 2 is an Upstream Regulator of CD4+ T Cells From Visceral Leishmaniasis Patients With Therapeutic Potential
Abstract
Control of visceral leishmaniasis (VL) caused by Leishmania donovani requires interferon-γ production by CD4+ T cells. In VL patients, antiparasitic CD4+ T-cell responses are ineffective for unknown reasons. In this study, we measured the expression of genes associated with various immune functions in these cells from VL patients and compared them to CD4+ T cells from the same patients after drug treatment and from endemic controls. We found reduced GATA3, RORC, and FOXP3 gene expression in CD4+ T cells of VL patients, associated with reduced Th2, Th17, and FOXP3+CD4+ T regulatory cell frequencies in VL patient blood. Interleukin 2 (IL-2) was an important upstream regulator of CD4+ T cells from VL patients, and functional studies demonstrated the therapeutic potential of IL-2 for improving antiparasitic immunity. Together, these results provide new insights into the characteristics of CD4+ T cells from VL patients that can be used to improve antiparasitic immune responses.
Keywords: Leishmania donovani; CD4+ T cells; IL-2; visceral leishmaniasis.
© The Author(s) 2019. Published by Oxford University Press for the Infectious Diseases Society of America. All rights reserved. For permissions, e-mail: journals.permissions@oup.com.
Figures
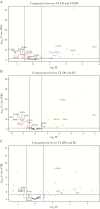


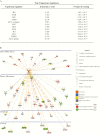
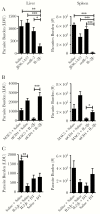
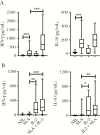
References
-
- Rosas LE, Snider HM, Barbi J, et al. . Cutting edge: STAT1 and T-bet play distinct roles in determining outcome of visceral leishmaniasis caused by Leishmania donovani. J Immunol 2006; 177:22–5. - PubMed
-
- Janeway CA Jr, Medzhitov R. Innate immune recognition. Annu Rev Immunol 2002; 20:197–216. - PubMed
-
- Sakaguchi S, Yamaguchi T, Nomura T, Ono M. Regulatory T cells and immune tolerance. Cell 2008; 133:775–87. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

