Fluid forces shape the embryonic heart: Insights from zebrafish
- PMID: 30797515
- PMCID: PMC6394863
- DOI: 10.1016/bs.ctdb.2018.12.009
Fluid forces shape the embryonic heart: Insights from zebrafish
Abstract
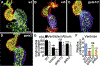
Heart formation involves a complex series of tissue rearrangements, during which regions of the developing organ expand, bend, converge, and protrude in order to create the specific shapes of important cardiac components. Much of this morphogenesis takes place while cardiac function is underway, with blood flowing through the rapidly contracting chambers. Fluid forces are therefore likely to influence the regulation of cardiac morphogenesis, but it is not yet clear how these biomechanical cues direct specific cellular behaviors. In recent years, the optical accessibility and genetic amenability of zebrafish embryos have facilitated unique opportunities to integrate the analysis of flow parameters with the molecular and cellular dynamics underlying cardiogenesis. Consequently, we are making progress toward a comprehensive view of the biomechanical regulation of cardiac chamber emergence, atrioventricular canal differentiation, and ventricular trabeculation. In this review, we highlight a series of studies in zebrafish that have provided new insight into how cardiac function can shape cardiac morphology, with a particular focus on how hemodynamics can impact cardiac cell behavior. Over the long-term, this knowledge will undoubtedly guide our consideration of the potential causes of congenital heart disease.
Keywords: Atrioventricular canal; Blood flow; Cardiac chambers; Heart development; Trabeculation.
© 2019 Elsevier Inc. All rights reserved.
Figures

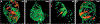

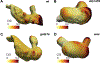

References
-
- Anton H, Harlepp S, Ramspacher C, Wu D, Monduc F, Bhat S, et al. (2013). Pulse Propagation by a Capacitive Mechanism Drives Embryonic Blood Flow. Development, 140, 4426–4434. - PubMed
-
- Bagatto B, Burggren W (2006). A Three-Dimensional Functional Assessment of Heart and Vessel Development in the Larva of the Zebrafish (Danio Rerio). Physiological and Biochemical Zoology, 79, 194–201. - PubMed
-
- Baratchi S, Khoshmanesh K, Woodman OL, Potocnik S, Peter K, McIntyre P (2017). Molecular Sensors of Blood Flow in Endothelial Cells. Trends in Molecular Medicine, 23, 850–868. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

