Extracellular signal-regulated kinases mediate the enhancing effects of inflammatory mediators on resurgent currents in dorsal root ganglion neurons
- PMID: 30803321
- PMCID: PMC6452589
- DOI: 10.1177/1744806919837104
Extracellular signal-regulated kinases mediate the enhancing effects of inflammatory mediators on resurgent currents in dorsal root ganglion neurons
Abstract
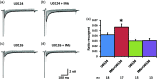
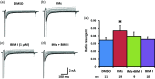
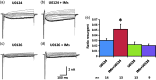
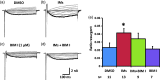
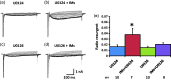
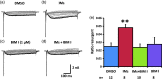
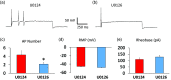
Previously we reported that a group of inflammatory mediators significantly enhanced resurgent currents in dorsal root ganglion neurons. To understand the underlying intracellular signaling mechanism, we investigated the effects of inhibition of extracellular signal-regulated kinases and protein kinase C on the enhancing effects of inflammatory mediators on resurgent currents in rat dorsal root ganglion neurons. We found that the extracellular signal-regulated kinases inhibitor U0126 completely prevented the enhancing effects of the inflammatory mediators on both Tetrodotoxin-sensitive and Tetrodotoxin-resistant resurgent currents in both small and medium dorsal root ganglion neurons. U0126 substantially reduced repetitive firing in small dorsal root ganglion neurons exposed to inflammatory mediators, consistent with prevention of resurgent current amplitude increases. The protein kinase C inhibitor Bisindolylmaleimide I also showed attenuating effects on resurgent currents, although to a lesser extent compared to extracellular signal-regulated kinases inhibition. These results indicate a critical role of extracellular signal-regulated kinases signaling in modulating resurgent currents and membrane excitability in dorsal root ganglion neurons treated with inflammatory mediators. It is also suggested that targeting extracellular signal-regulated kinases-resurgent currents might be a useful strategy to reduce inflammatory pain.
Keywords: Resurgent currents; Tetrodotoxin-resistant; Tetrodotoxin-sensitive; dorsal root ganglion; extracellular signal-regulated kinases; inflammatory mediators; protein kinase C.
Figures
Similar articles
-
Tetrodotoxin-resistant sodium channels in sensory neurons generate slow resurgent currents that are enhanced by inflammatory mediators.J Neurosci. 2014 May 21;34(21):7190-7. doi: 10.1523/JNEUROSCI.5011-13.2014. J Neurosci. 2014. PMID: 24849353 Free PMC article.
-
PGE2 modulates the tetrodotoxin-resistant sodium current in neonatal rat dorsal root ganglion neurones via the cyclic AMP-protein kinase A cascade.J Physiol. 1996 Sep 1;495 ( Pt 2)(Pt 2):429-40. doi: 10.1113/jphysiol.1996.sp021604. J Physiol. 1996. PMID: 8887754 Free PMC article.
-
FHF2 isoforms differentially regulate Nav1.6-mediated resurgent sodium currents in dorsal root ganglion neurons.Pflugers Arch. 2017 Feb;469(2):195-212. doi: 10.1007/s00424-016-1911-9. Epub 2016 Dec 20. Pflugers Arch. 2017. PMID: 27999940 Free PMC article.
-
Single-channel analysis of tetrodotoxin-sensitive and tetrodotoxin-resistant sodium channels in rat dorsal root ganglion neurons.Brain Res. 1994 Jul 11;650(2):341-6. doi: 10.1016/0006-8993(94)91803-1. Brain Res. 1994. PMID: 7953703
-
Phosphorylation of extracellular signal-related protein kinase is required for rapid facilitation of heat-induced currents in rat dorsal root ganglion neurons.Neuroscience. 2006 Nov 17;143(1):253-63. doi: 10.1016/j.neuroscience.2006.07.047. Epub 2006 Sep 14. Neuroscience. 2006. PMID: 16973292
Cited by
-
Oxaliplatin Depolarizes the IB4- Dorsal Root Ganglion Neurons to Drive the Development of Neuropathic Pain Through TRPM8 in Mice.Front Mol Neurosci. 2021 Jun 4;14:690858. doi: 10.3389/fnmol.2021.690858. eCollection 2021. Front Mol Neurosci. 2021. PMID: 34149356 Free PMC article.
-
Role of Intermediate Filaments in Blood-Brain Barrier in Health and Disease.Cells. 2021 Jun 5;10(6):1400. doi: 10.3390/cells10061400. Cells. 2021. PMID: 34198868 Free PMC article. Review.
-
Ojeok-san ameliorates visceral and somatic nociception in a mouse model of colitis induced colorectal cancer.PLoS One. 2022 Jun 23;17(6):e0270338. doi: 10.1371/journal.pone.0270338. eCollection 2022. PLoS One. 2022. PMID: 35737651 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

