Pentoxifylline Enhances the Apoptotic Effect of Carboplatin in Y79 Retinoblastoma Cells
- PMID: 30804118
- PMCID: PMC6506323
- DOI: 10.21873/invivo.11487
Pentoxifylline Enhances the Apoptotic Effect of Carboplatin in Y79 Retinoblastoma Cells
Abstract
Background/aim: Retinoblastoma (RB) is the most common primary intraocular malignancy. Carboplatin (CPt) is a DNA damage-inducing agent that is widely used for the treatment of RB. Unfortunately, this drug also activates the transcription factor nuclear factor-kappa B (NF-ĸB), leading to promotion of tumor survival. Pentoxifylline (PTX) is a drug that inhibits the phosphorylation of I kappa B-alpha (IĸBα) in serines 32 and 36, and this disrupts NF-ĸB activity that promotes tumor survival. The goal of this study was to evaluate the effect of the PTX on the antitumor activity of CPt.
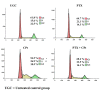
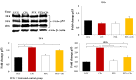
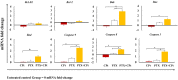
Materials and methods: Y79 RB cells were treated with CPt, PTX, or both. Cell viability, apoptosis, loss of mitochondrial membrane potential, the activity of caspase-9, -8, and -3, cytochrome c release, cell-cycle progression, p53, and phosphorylation of IĸBα, and pro- and anti-apoptotic genes were evaluated.
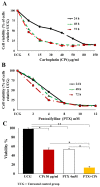
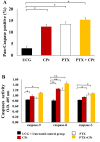
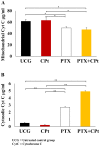
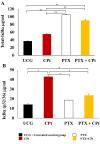
Results: Both drugs significantly affected the viability of the Y79 RB cells in a time- and dose-dependent manner. The PTX+CPt combination exhibited the highest rate of apoptosis, a decrease in cell viability and significant caspase activation, as well as loss of mitochondrial membrane potential, release of cytochrome c, and increased p53 protein levels. Cells treated with PTX alone displayed decreased I kappa B-alpha phosphorylation, compared to the CPt treated group. In addition, the PTX+CPt combination treatment induced up-regulation of the proapoptotic genes Bax, Bad, Bak, and caspases- 3, -8, and -9, compared to the CPt and PTX individual treated groups.
Conclusion: PTX induces apoptosis per se and increases the CPt-induced apoptosis, augmenting its antitumor effectiveness.
Keywords: NF-ĸB; Pentoxifylline; Y79 cells; apoptosis; carboplatin.
Copyright© 2019, International Institute of Anticancer Research (Dr. George J. Delinasios), All rights reserved.
Conflict of interest statement
The Authors declare that they have no conflicting interests.
Figures
Similar articles
-
Sensitization of cervix cancer cells to Adriamycin by Pentoxifylline induces an increase in apoptosis and decrease senescence.Mol Cancer. 2010 May 19;9:114. doi: 10.1186/1476-4598-9-114. Mol Cancer. 2010. PMID: 20482878 Free PMC article.
-
Paclitaxel and beta-lapachone synergistically induce apoptosis in human retinoblastoma Y79 cells by downregulating the levels of phospho-Akt.J Cell Physiol. 2010 Feb;222(2):433-43. doi: 10.1002/jcp.21983. J Cell Physiol. 2010. PMID: 19918798
-
Pentoxifylline and the proteasome inhibitor MG132 induce apoptosis in human leukemia U937 cells through a decrease in the expression of Bcl-2 and Bcl-XL and phosphorylation of p65.J Biomed Sci. 2013 Feb 28;20(1):13. doi: 10.1186/1423-0127-20-13. J Biomed Sci. 2013. PMID: 23445492 Free PMC article.
-
Low doses of paclitaxel potently induce apoptosis in human retinoblastoma Y79 cells by up-regulating E2F1.Int J Oncol. 2008 Oct;33(4):677-87. Int J Oncol. 2008. PMID: 18813780
-
Chemotherapy with a molecular rational basis, pentoxifylline as a promising antitumor drug.Ann Med Surg (Lond). 2025 Feb 28;87(3):1506-1528. doi: 10.1097/MS9.0000000000003043. eCollection 2025 Mar. Ann Med Surg (Lond). 2025. PMID: 40213176 Free PMC article. Review.
Cited by
-
Role of Mitochondria and Lysosomes in the Selective Cytotoxicity of Cold Atmospheric Plasma on Retinoblastoma Cells.Iran J Pharm Res. 2020 Fall;19(4):203-215. doi: 10.22037/ijpr.2020.114165.14703. Iran J Pharm Res. 2020. PMID: 33841536 Free PMC article.
-
Pentoxifylline Sensitizes Cisplatin-Resistant Human Cervical Cancer Cells to Cisplatin Treatment: Involvement of Mitochondrial and NF-Kappa B Pathways.Front Oncol. 2020 Dec 16;10:592706. doi: 10.3389/fonc.2020.592706. eCollection 2020. Front Oncol. 2020. PMID: 33680921 Free PMC article.
-
Carboplatin Inhibits the Progression of Retinoblastoma Through IncRNA XIST/miR-200a-3p/NRP1 Axis.Drug Des Devel Ther. 2020 Aug 21;14:3417-3427. doi: 10.2147/DDDT.S256813. eCollection 2020. Drug Des Devel Ther. 2020. PMID: 32904674 Free PMC article.
-
Sensitizing the cytotoxic action of Docetaxel induced by Pentoxifylline in a PC3 prostate cancer cell line.BMC Urol. 2021 Mar 12;21(1):38. doi: 10.1186/s12894-021-00807-6. BMC Urol. 2021. PMID: 33711972 Free PMC article.
-
Expression of ornithine decarboxylase in peripheral blood mononuclear cells from patients with pancreatic adenocarcinoma: A preliminary report.Biomed Rep. 2024 Jan 17;20(3):38. doi: 10.3892/br.2024.1726. eCollection 2024 Mar. Biomed Rep. 2024. PMID: 38343658 Free PMC article.
References
-
- Bartuma K, Pal N, Kosek S, Holm S, All-Ericsson C. A 10-year experience of outcome in chemotherapy-treated hereditary retinoblastoma. Acta Ophthalmol. 2014;92(5):404–411. - PubMed
-
- Rebucci M, Michiels C. Molecular aspects of cancer cell resistance to chemotherapy. Biochem Pharmacol. 2013;85(9):1219–1226. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
