Revisiting Anti-tuberculosis Therapeutic Strategies That Target the Peptidoglycan Structure and Synthesis
- PMID: 30804921
- PMCID: PMC6378297
- DOI: 10.3389/fmicb.2019.00190
Revisiting Anti-tuberculosis Therapeutic Strategies That Target the Peptidoglycan Structure and Synthesis
Abstract
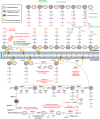
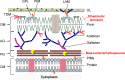
Tuberculosis (TB), which is caused by Mycobacterium tuberculosis (Mtb), is one of the leading cause of death by an infectious diseases. The biosynthesis of the mycobacterial cell wall (CW) is an area of increasing research significance, as numerous antibiotics used to treat TB target biosynthesis pathways of essential CW components. The main feature of the mycobacterial cell envelope is an intricate structure, the mycolyl-arabinogalactan-peptidoglycan (mAGP) complex responsible for its innate resistance to many commonly used antibiotics and involved in virulence. A hallmark of mAGP is its unusual peptidoglycan (PG) layer, which has subtleties that play a key role in virulence by enabling pathogenic species to survive inside the host and resist antibiotic pressure. This dynamic and essential structure is not a target of currently used therapeutics as Mtb is considered naturally resistant to most β-lactam antibiotics due to a highly active β-lactamase (BlaC) that efficiently hydrolyses many β-lactam drugs to render them ineffective. The emergence of multidrug- and extensive drug-resistant strains to the available antibiotics has become a serious health threat, places an immense burden on health care systems, and poses particular therapeutic challenges. Therefore, it is crucial to explore additional Mtb vulnerabilities that can be used to combat TB. Remodeling PG enzymes that catalyze biosynthesis and recycling of the PG are essential to the viability of Mtb and are therefore attractive targets for novel antibiotics research. This article reviews PG as an alternative antibiotic target for TB treatment, how Mtb has developed resistance to currently available antibiotics directed to PG biosynthesis, and the potential of targeting this essential structure to tackle TB by attacking alternative enzymatic activities involved in Mtb PG modifications and metabolism.
Keywords: antibiotic resistance; cell wall; mycobacteria; mycobacteriophage lysis enzymes; peptidoglycan; tuberculosis; β-lactams.
Figures
Similar articles
-
Identification of drivers of mycobacterial resistance to peptidoglycan synthesis inhibitors.Front Microbiol. 2022 Sep 6;13:985871. doi: 10.3389/fmicb.2022.985871. eCollection 2022. Front Microbiol. 2022. PMID: 36147841 Free PMC article.
-
CRISPRi-mediated characterization of novel anti-tuberculosis targets: Mycobacterial peptidoglycan modifications promote beta-lactam resistance and intracellular survival.Front Cell Infect Microbiol. 2023 Mar 15;13:1089911. doi: 10.3389/fcimb.2023.1089911. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37009497 Free PMC article.
-
Editorial: Current status and perspective on drug targets in tubercle bacilli and drug design of antituberculous agents based on structure-activity relationship.Curr Pharm Des. 2014;20(27):4305-6. doi: 10.2174/1381612819666131118203915. Curr Pharm Des. 2014. PMID: 24245755
-
Mycobacterial cell wall biosynthesis: a multifaceted antibiotic target.Parasitology. 2018 Feb;145(2):116-133. doi: 10.1017/S0031182016002377. Epub 2016 Dec 15. Parasitology. 2018. PMID: 27976597 Free PMC article. Review.
-
Structure, function and biosynthesis of the Mycobacterium tuberculosis cell wall: arabinogalactan and lipoarabinomannan assembly with a view to discovering new drug targets.Biochem Soc Trans. 2007 Nov;35(Pt 5):1325-8. doi: 10.1042/BST0351325. Biochem Soc Trans. 2007. PMID: 17956343 Review.
Cited by
-
Acyldepsipeptide Analogues: A Future Generation Antibiotics for Tuberculosis Treatment.Pharmaceutics. 2022 Sep 15;14(9):1956. doi: 10.3390/pharmaceutics14091956. Pharmaceutics. 2022. PMID: 36145704 Free PMC article. Review.
-
Construction and Use of Transposon MycoTetOP 2 for Isolation of Conditional Mycobacteria Mutants.Front Microbiol. 2020 Jan 21;10:3091. doi: 10.3389/fmicb.2019.03091. eCollection 2019. Front Microbiol. 2020. PMID: 32038540 Free PMC article.
-
Identification of drivers of mycobacterial resistance to peptidoglycan synthesis inhibitors.Front Microbiol. 2022 Sep 6;13:985871. doi: 10.3389/fmicb.2022.985871. eCollection 2022. Front Microbiol. 2022. PMID: 36147841 Free PMC article.
-
Ethambutol and meropenem/clavulanate synergy promotes enhanced extracellular and intracellular killing of Mycobacterium tuberculosis.Antimicrob Agents Chemother. 2024 Apr 3;68(4):e0158623. doi: 10.1128/aac.01586-23. Epub 2024 Feb 27. Antimicrob Agents Chemother. 2024. PMID: 38411952 Free PMC article.
-
Bacteriophages of Mycobacterium tuberculosis, their diversity, and potential therapeutic uses: a review.BMC Infect Dis. 2022 Dec 22;22(1):957. doi: 10.1186/s12879-022-07944-9. BMC Infect Dis. 2022. PMID: 36550444 Free PMC article. Review.
References
-
- Atilano M. L., Yates J., Glittenberg M., Filipe S. R., Ligoxygakis P. (2011). Wall teichoic acids of Staphylococcus aureus limit recognition by the Drosophila peptidoglycan recognition protein-SA to promote pathogenicity. PLoS Pathog. 7:e1002421. 10.1371/journal.ppat.1002421, PMID: - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

