Ferritin Light Chain Confers Protection Against Sepsis-Induced Inflammation and Organ Injury
- PMID: 30804939
- PMCID: PMC6371952
- DOI: 10.3389/fimmu.2019.00131
Ferritin Light Chain Confers Protection Against Sepsis-Induced Inflammation and Organ Injury
Abstract
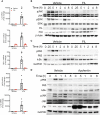
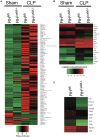
Despite the prevalence and recognition of its detrimental impact, clinical complications of sepsis remain a major challenge. Here, we investigated the effects of myeloid ferritin heavy chain (FtH) in regulating the pathogenic sequelae of sepsis. We demonstrate that deletion of myeloid FtH leads to protection against lipopolysaccharide-induced endotoxemia and cecal ligation and puncture (CLP)-induced model of sepsis as evidenced by reduced cytokine levels, multi-organ dysfunction and mortality. We identified that such protection is predominantly mediated by the compensatory increase in circulating ferritin (ferritin light chain; FtL) in the absence of myeloid FtH. Our in vitro and in vivo studies indicate that prior exposure to ferritin light chain restrains an otherwise dysregulated response to infection. These findings are mediated by an inhibitory action of FtL on NF-κB activation, a key signaling pathway that is implicated in the pathogenesis of sepsis. We further identified that LPS mediated activation of MAPK pathways, specifically, JNK, and ERK were also reduced with FtL pre-treatment. Taken together, our findings elucidate a crucial immunomodulatory function for circulating ferritin that challenges the traditional view of this protein as a mere marker of body iron stores. Accordingly, these findings will stimulate investigations to the adaptive nature of this protein in diverse clinical settings.
Keywords: LPS; NF-κB; cytokine; ferritin; inflammatory response; multi-organ injury; myeloid cells; sepsis.
Figures





Similar articles
-
Myeloid-specific ferritin light chain deletion does not exacerbate sepsis-associated AKI.Am J Physiol Renal Physiol. 2024 Jul 1;327(1):F171-F183. doi: 10.1152/ajprenal.00043.2024. Epub 2024 May 23. Am J Physiol Renal Physiol. 2024. PMID: 38779751 Free PMC article.
-
LPS pretreatment ameliorates multiple organ injuries and improves survival in a murine model of polymicrobial sepsis.Inflamm Res. 2011 Sep;60(9):841-9. doi: 10.1007/s00011-011-0342-5. Epub 2011 May 10. Inflamm Res. 2011. PMID: 21556916
-
Ketamine inhibits LPS-induced HGMB1 release in vitro and in vivo.Int Immunopharmacol. 2014 Nov;23(1):14-26. doi: 10.1016/j.intimp.2014.08.003. Epub 2014 Aug 13. Int Immunopharmacol. 2014. PMID: 25133650
-
Epinecidin-1 protects mice from LPS-induced endotoxemia and cecal ligation and puncture-induced polymicrobial sepsis.Biochim Biophys Acta Mol Basis Dis. 2017 Dec;1863(12):3028-3037. doi: 10.1016/j.bbadis.2017.08.032. Epub 2017 Sep 4. Biochim Biophys Acta Mol Basis Dis. 2017. PMID: 28882626
-
Fisetin alleviates sepsis-induced multiple organ dysfunction in mice via inhibiting p38 MAPK/MK2 signaling.Acta Pharmacol Sin. 2020 Oct;41(10):1348-1356. doi: 10.1038/s41401-020-0462-y. Epub 2020 Jul 13. Acta Pharmacol Sin. 2020. PMID: 32661350 Free PMC article.
Cited by
-
Pro-inflammatory properties of H-ferritin on human macrophages, ex vivo and in vitro observations.Sci Rep. 2020 Jul 22;10(1):12232. doi: 10.1038/s41598-020-69031-w. Sci Rep. 2020. PMID: 32699419 Free PMC article.
-
The joint involvement in adult onset Still's disease is characterised by a peculiar magnetic resonance imaging and a specific transcriptomic profile.Sci Rep. 2021 Jun 14;11(1):12455. doi: 10.1038/s41598-021-91613-5. Sci Rep. 2021. PMID: 34127696 Free PMC article.
-
Mechanisms controlling cellular and systemic iron homeostasis.Nat Rev Mol Cell Biol. 2024 Feb;25(2):133-155. doi: 10.1038/s41580-023-00648-1. Epub 2023 Oct 2. Nat Rev Mol Cell Biol. 2024. PMID: 37783783 Review.
-
A Macrophage-Related Gene Signature for Identifying COPD Based on Bioinformatics and ex vivo Experiments.J Inflamm Res. 2023 Nov 29;16:5647-5665. doi: 10.2147/JIR.S438308. eCollection 2023. J Inflamm Res. 2023. PMID: 38050560 Free PMC article.
-
Puerarin@Chitosan composite for infected bone repair through mimicking the bio-functions of antimicrobial peptides.Bioact Mater. 2022 Sep 20;21:520-530. doi: 10.1016/j.bioactmat.2022.09.005. eCollection 2023 Mar. Bioact Mater. 2022. PMID: 36185735 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

