LncRNA IMFNCR Promotes Intramuscular Adipocyte Differentiation by Sponging miR-128-3p and miR-27b-3p
- PMID: 30804984
- PMCID: PMC6378276
- DOI: 10.3389/fgene.2019.00042
LncRNA IMFNCR Promotes Intramuscular Adipocyte Differentiation by Sponging miR-128-3p and miR-27b-3p
Abstract
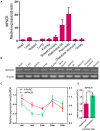
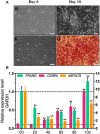
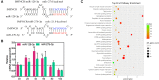
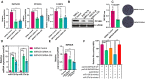
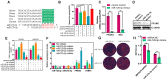
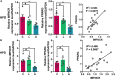
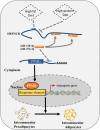
Poultry meat quality is affected by many factors, among which intramuscular fat (IMF) is predominant. IMF content affects the tenderness, juiciness, and flavor of chicken. An increasing number of studies are focusing on the functions of lncRNAs in adipocyte differentiation. However, little is known about lncRNAs associated with intramuscular adipocyte differentiation. In the present study, we focused on an up-regulated lncRNA during intramuscular adipogenetic differentiation, which we named intramuscular fat-associated long non-coding RNA (IMFNCR). IMFNCR promotes intramuscular adipocyte differentiation. In-depth analyses showed that IMFNCR acts as a molecular sponge for miR-128-3p and miR-27b-3p and that PPARG is a direct target of miR-128-3p and miR-27b-3p in chicken. High-fat and high-protein diet inhibited chicken IMFNCR level in vivo. Moreover, IMFNCR level was positively correlated with PPARG mRNA level in chicken breast muscle tissues, a vital corollary to ceRNA function. Altogether, our research showed that IMFNCR acts as a ceRNA to sequester miR-128-3p and miR-27b-3p, leading to heightened PPARG expression, and thus promotes intramuscular adipocyte differentiation. Taken together, our findings may contribute to a more thorough understanding of chicken IMF deposition and the improvement of poultry meat quality.
Keywords: ceRNA; chicken; intramuscular adipocyte differentiation; long non-coding RNA; miRNA.
Figures

Similar articles
-
Integrated Analysis of MiRNA and Genes Associated with Meat Quality Reveals that Gga-MiR-140-5p Affects Intramuscular Fat Deposition in Chickens.Cell Physiol Biochem. 2018;46(6):2421-2433. doi: 10.1159/000489649. Epub 2018 May 4. Cell Physiol Biochem. 2018. PMID: 29742492
-
gga-miRNA-18b-3p Inhibits Intramuscular Adipocytes Differentiation in Chicken by Targeting the ACOT13 Gene.Cells. 2019 Jun 7;8(6):556. doi: 10.3390/cells8060556. Cells. 2019. PMID: 31181634 Free PMC article.
-
miR-128-3p inhibits intramuscular adipocytes differentiation in chickens by downregulating FDPS.BMC Genomics. 2023 Sep 12;24(1):540. doi: 10.1186/s12864-023-09649-y. BMC Genomics. 2023. PMID: 37700222 Free PMC article.
-
miRNA-223 targets the GPAM gene and regulates the differentiation of intramuscular adipocytes.Gene. 2019 Feb 15;685:106-113. doi: 10.1016/j.gene.2018.10.054. Epub 2018 Oct 30. Gene. 2019. PMID: 30389563
-
Genomic Insights into Molecular Regulation Mechanisms of Intramuscular Fat Deposition in Chicken.Genes (Basel). 2023 Dec 10;14(12):2197. doi: 10.3390/genes14122197. Genes (Basel). 2023. PMID: 38137019 Free PMC article. Review.
Cited by
-
circFLT1 and lncCCPG1 Sponges miR-93 to Regulate the Proliferation and Differentiation of Adipocytes by Promoting lncSLC30A9 Expression.Mol Ther Nucleic Acids. 2020 Sep 16;22:484-499. doi: 10.1016/j.omtn.2020.09.011. eCollection 2020 Dec 4. Mol Ther Nucleic Acids. 2020. PMID: 33230451 Free PMC article.
-
White Adipocyte Stem Cell Expansion Through Infant Formula Feeding: New Insights into Epigenetic Programming Explaining the Early Protein Hypothesis of Obesity.Int J Mol Sci. 2025 May 8;26(10):4493. doi: 10.3390/ijms26104493. Int J Mol Sci. 2025. PMID: 40429638 Free PMC article. Review.
-
Proteome Analysis Related to Unsaturated Fatty Acid Synthesis by Interfering with Bovine Adipocyte ACSL1 Gene.Antioxidants (Basel). 2024 May 24;13(6):641. doi: 10.3390/antiox13060641. Antioxidants (Basel). 2024. PMID: 38929080 Free PMC article.
-
Resveratrol Inhibits Proliferation and Differentiation of Porcine Preadipocytes by a Novel LincRNA-ROFM/miR-133b/AdipoQ Pathway.Foods. 2022 Sep 3;11(17):2690. doi: 10.3390/foods11172690. Foods. 2022. PMID: 36076875 Free PMC article.
-
Noncoding RNA profiling in omentum adipose tissue from obese patients and the identification of novel metabolic biomarkers.Front Genet. 2025 Feb 6;16:1533637. doi: 10.3389/fgene.2025.1533637. eCollection 2025. Front Genet. 2025. PMID: 39981261 Free PMC article.
References
-
- Aaslyng M. D., Bejerholm C., Ertbjerg P., Bertram H. C., Andersen H. J. (2003). Cooking loss and juiciness of pork in relation to raw meat quality and cooking procedure. Food Q. Prefer. 14 277–288. 10.1016/S0950-3293(02)00086-1 - DOI
-
- Bai P., Houten S. M., Huber A., Schreiber V., Watanabe M., Kiss B., et al. (2007). PARP-2 controls adipocyte differentiation and adipose tissue function through the regulation of the activity of the RXR/PPARgamma heterodimer. Exp. Eye Res. 17 765–771. - PubMed
LinkOut - more resources
Full Text Sources

