A tri state mechanism for oxygen release in fish hemoglobin: Using Barbus sharpeyi as a model
- PMID: 30805377
- PMCID: PMC6373571
A tri state mechanism for oxygen release in fish hemoglobin: Using Barbus sharpeyi as a model
Abstract
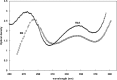
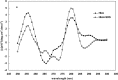
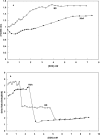
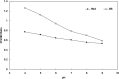
Hemoglobin is a porphyrin containing protein with an α2β2 tetrameric structure and like other porphyrin compounds shows spectral behavior of species specific characteristics. Researchers tend to relate bands in the hemoglobin spectra to certain structural and/or functional features. Given the fact th↔at hemoglobin is the main oxygen carrier in animals functioning through the Oxy↔Deoxy equilibrium, the determination of oxy and deoxy conformations of hemoglobins of different animals may shed light on their oxygen binding properties. Absorption spectra at 280 and 373nm have been widely used to quantitate the formation of hemoglobin deoxy conformation. In the present work, however, we used an optical density ratio of OD373/OD280 as an index for deoxy formation. This ratio was determined for Barbus sharpeyi and human hemoglobins at different SDS concentrations, pH levels and temperatures to compare them from a structure-function point of view. Our data showed that under low concentrations of SDS (<2mM) Barbus sharpeyi hemoglobin folds in a tri-state pattern while human hemoglobin folds through a two-state phenomenon. This finding indicates that in contrast to those of other non aquatic animals, the hemoglobin of Barbus sharpeyi has a loosely folded tetrameric structure with remarkably more oxygen affinity.
Keywords: Barbus sharpeyi; Hemoglobin; Tri State Mechanism.
Figures


Similar articles
-
Comparison of human and shirbot (Cyprinidae: Barbus grypus) hemoglobin: a structure-function prospective.Protein Pept Lett. 2011 Nov;18(11):1072-7. doi: 10.2174/092986611797200904. Protein Pept Lett. 2011. PMID: 21675952
-
T-quaternary structure of oxy human adult hemoglobin in the presence of two allosteric effectors, L35 and IHP.Biochim Biophys Acta. 2011 Oct;1807(10):1253-61. doi: 10.1016/j.bbabio.2011.06.004. Epub 2011 Jun 15. Biochim Biophys Acta. 2011. PMID: 21703224
-
Ruthenium-iron hybrid hemoglobins as a model for partially liganded hemoglobin: oxygen equilibrium curves and resonance Raman spectra.Biochemistry. 1989 Oct 17;28(21):8603-9. doi: 10.1021/bi00447a050. Biochemistry. 1989. PMID: 2605210
-
Hemoglobin function in the vertebrates: an evolutionary model.J Mol Evol. 1975 Dec 29;6(4):285-307. doi: 10.1007/BF01794636. J Mol Evol. 1975. PMID: 1543 Review.
-
Mutagenic dissection of hemoglobin cooperativity: effects of amino acid alteration on subunit assembly of oxy and deoxy tetramers.Proteins. 1992 Nov;14(3):333-50. doi: 10.1002/prot.340140303. Proteins. 1992. PMID: 1438173 Review.
References
-
- Nelson L, Cox M. Lehinger Principles of Biochemistry. 4th Ed. New York: Freeman WH and Company; 2005. Protein Function.
-
- Vasirev AS, Luk'yanenko VI. Spectrophotometrie parameters of conformational states of hemoglobin and its complexes with ligands in the sturgeons. J Evol Biochem Physiol. 2000;36:5–10.
-
- Nagatomo S, Jin Y, Nagai M, Hori H, Kitagawa T. Changes in the abnormal a- subunit upon CO-binding to the normal b-subunit of Hb M Boston: resonance Raman, EPR and CD study. Biophys Chem. 2002;98:217–232. - PubMed
-
- Reinot T, Hayes JM, Small GJ, Zerner MC. Q-band splitting and relaxation of aluminum phthalocyanine tetrasulfonate. Chem Phys Lett. 1999;299:410–416.
-
- Eaton WA, Hofrichter J. Polarized absorption and linear dichroism spectroscopy of hemoglobin. Methods Enzymol. 1981;76:175–261. - PubMed
LinkOut - more resources
Full Text Sources
