Myosin-X Silencing in the Trabecular Meshwork Suggests a Role for Tunneling Nanotubes in Outflow Regulation
- PMID: 30807639
- PMCID: PMC6390986
- DOI: 10.1167/iovs.18-26055
Myosin-X Silencing in the Trabecular Meshwork Suggests a Role for Tunneling Nanotubes in Outflow Regulation
Abstract
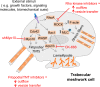
Purpose: The actin cytoskeleton plays a key role in outflow regulation through the trabecular meshwork (TM). Although actin stress fibers are a target of glaucoma therapies, the role of other actin cellular structures is unclear. Myosin-X (Myo10) is an actin-binding protein that is involved in tunneling nanotube (TNT) and filopodia formation. Here, we inhibited Myo10 pharmacologically or by gene silencing to investigate the role of filopodia/TNTs in the TM.
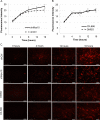
Methods: Short hairpin RNA interference (RNAi) silencing lentivirus targeting myosin-X (shMyo10) was generated. Human anterior segments were perfused with shMyo10 or CK-666, an Arp2/3 inhibitor. Confocal microscopy investigated the colocalization of Myo10 with matrix metalloproteinase (MMPs). Western immunoblotting investigated the protein levels of MMPs and extracellular matrix (ECM) proteins. MMP activity and phagocytosis assays were performed.
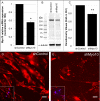
Results: CK-666 and shMyo10-silencing lentivirus caused a significant reduction in outflow rates in anterior segment perfusion culture, an ex vivo method to study intraocular pressure regulation. In human TM cells, Myo10 colocalized with MMP2, MMP14, and cortactin in podosome-like structures, which function as regions of focal ECM degradation. Furthermore, MMP activity, thrombospondin-1 and SPARC protein levels were significantly reduced in the media of CK-666-treated and shMyo10-silenced TM cells. However, neither Myo10 silencing or CK-666 treatment significantly affected phagocytic uptake.
Conclusions: Inhibiting filopodia/TNTs caused opposite effects on outflow compared with inhibiting stress fibers. Moreover, Myo10 may also play a role in focal ECM degradation in TM cells. Our results provide additional insight into the function of actin supramolecular assemblies and actin-binding proteins in outflow regulation.
Figures



References
-
- Quigley HA. Glaucoma. Lancet. 2011;377:1367–1377. - PubMed
-
- Nobes CD, Rho Hall A. rac and cdc42 GTPases: regulators of actin structures, cell adhesion and motility. Biochem Soc Trans. 1995;23:456–459. - PubMed
-
- Rao PV, Deng PF, Kumar J, Epstein DL. Modulation of aqueous humor outflow facility by the Rho kinase-specific inhibitor Y-27632. Invest Ophthalmol Vis Sci. 2001;42:1029–1037. - PubMed
-
- Vittitow JL, Garg R, Rowlette LL, Epstein DL, O'Brien ET, Borras T. Gene transfer of dominant-negative RhoA increases outflow facility in perfused human anterior segment cultures. Mol Vis. 2002;8:32–44. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

